The relation between enthalpy h, stagnation enthalpy h0, and kinetic energy ![]() is:
is:

For an ideal gas with constant specific heat, Eq. (6.15) can be written as:
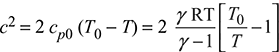
Since ![]() = γRT, where γ = cp0/cv0
= γRT, where γ = cp0/cv0
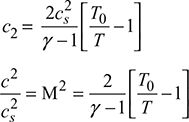

For an isentropic process,
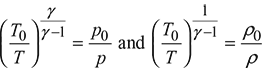
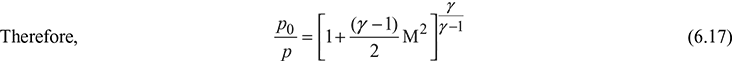

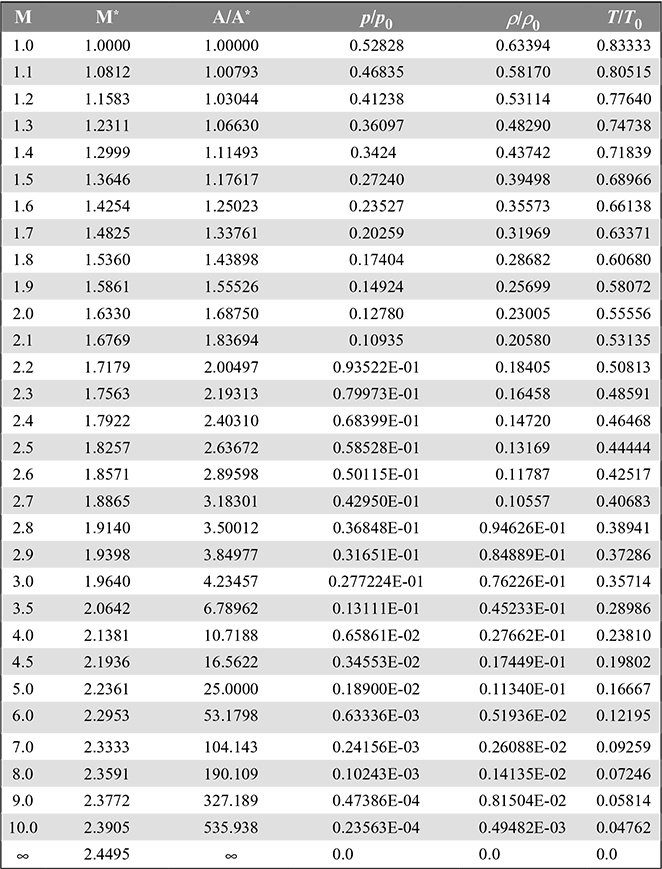
Values of ![]() are given as a function of M in Table 6.1 for the value of γ = 1.40.
are given as a function of M in Table 6.1 for the value of γ = 1.40.
Table 6.1 One-dimensional insentropic compressible-flow functions for an ideal gas with constant specific heat and molecular weight and γ = 1.4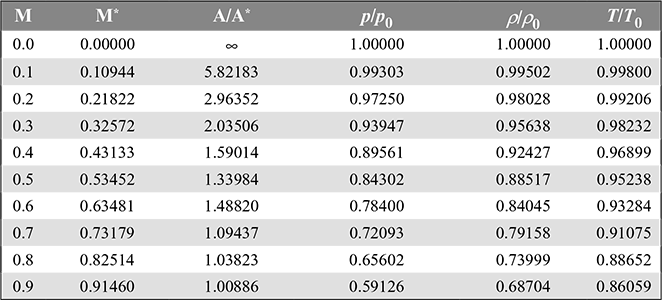
The conditions at the throat of the nozzle can be found by putting M = 1 at the throat. The properties at the throat are denoted as an asterisk (*) and are referred to as critical properties.
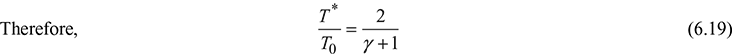


Table 6.2 gives these ratios for various values of k.
Table 6.2 Critical properties for isentropic flow of an ideal gas
Leave a Reply