- Enthalpy or total heat of water: It is defined as the quantity of heat required to raise the temperature of 1 kg of water from 0°C to its boiling point or saturation temperature corresponding to the pressure applied. It is also called enthalpy of saturated water or liquid heat and is represented by hf.hf = specific heat of water, cpw × rise in temperature= 4.187 × Δt, kJ/kgNote that enthalpy of saturated water can also be found from the steam table based on saturation temperature (Appendix A.1.1).
- Heat of steam or latent heat of vapourisation: Latent heat of steam at a particular pressure may be defined as the quantity of heat required to convert 1 kg of water at its boiling point into dry saturated steam at the same pressure. It is denoted by hfg.
- Dryness fraction or quality: Dryness fraction of a liquid−vapour mixture is defined as the ratio of mass of dry steam actually present to the mass of the total mixture. It is denoted by x.Let ms = mass of dry steam (saturated vapour), kgmw = mass of water vapour in steam (saturated liquid), kgx = dryness fraction of the sample
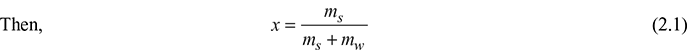 For dry steam, mw = 0 and x = 1.The quality of steam is the dryness fraction expressed as a percentage.Wetness fraction = 1 − x
For dry steam, mw = 0 and x = 1.The quality of steam is the dryness fraction expressed as a percentage.Wetness fraction = 1 − x - Specific volume of wet steam: The specific volume of steam is the volume of 1 kg of dry steam.Let V be the total volume of the liquid-vapour mixture, Vf be the volume of the saturated liquid and Vg be the volume of the saturated vapour.Total volume of liquid and vapour can be expressed in terms of their specific volume asVf = mwvfVg = msvgwhere vf and vg are the specific volumes of saturated liquid and saturated vapour, respectively.Specific volume of the mixture is then
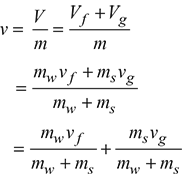 = (1 − x)v f + xvg= v f + x(vg − v f)
= (1 − x)v f + xvg= v f + x(vg − v f) Now vf << vg
Now vf << vg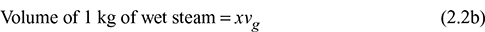
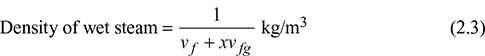
- Enthalpy of wet steam: It may be defined as the quantity of heat required to convert 1 kg of water at 0°C, at constant pressure, into wet steam. Similar to the analysis of specific volume, one can write
 where hf is the specific enthalpy of saturated water and hfg is the change in specific enthalpy during vapourisation. When steam is dry saturated, then x = 1
where hf is the specific enthalpy of saturated water and hfg is the change in specific enthalpy during vapourisation. When steam is dry saturated, then x = 1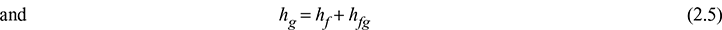
- Total enthalpy of superheated steam:Let cps = specific heat of superheated steamts = temperature of formation of steam, °Ctsup = temperature of superheated steam, °CThen heat of superheat = cps (tsup − ts)

 Note that enthalpy of superheated steam can also be found from the steam table (Appendix A.1.3).
Note that enthalpy of superheated steam can also be found from the steam table (Appendix A.1.3). - Internal energy of steam: The internal energy of steam may be defined as the actual heat energy stored in steam above the freezing point of water. It is the difference between the total heat of steam and the external work of evaporation. Similar to the analysis of specific volume, one can write for 1 kg of wet steam,
 where uf is the specific internal energy of saturated water and ufg is the change in specific internal energy during evaporation. From the known value of enthalpy, internal energy can be found as follows: uwet = h − pv1
where uf is the specific internal energy of saturated water and ufg is the change in specific internal energy during evaporation. From the known value of enthalpy, internal energy can be found as follows: uwet = h − pv1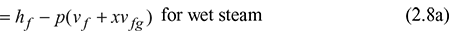 For superheated steam,hsup = hg + cps (tsup − ts)usup = hsup − pvsup
For superheated steam,hsup = hg + cps (tsup − ts)usup = hsup − pvsup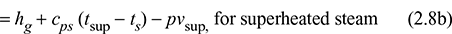
- Entropy of water: Consider 1 kg of water to be heated from a temperature Ts to T at a constant pressure.Then, change of entropy =
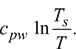 where cpw = specific heat of waterThe entropy of water is always reckoned above the freezing point of water.Therefore, Ts = saturation temperature of formation of steamT = absolute temperature of freezing water = 273 KEntropy of saturated water,
where cpw = specific heat of waterThe entropy of water is always reckoned above the freezing point of water.Therefore, Ts = saturation temperature of formation of steamT = absolute temperature of freezing water = 273 KEntropy of saturated water,
- Entropy of wet steam: Consider 1 kg of wet steam at an absolute pressure p bar.Let x = dryness fraction of steamTs = temperature of formation of steamsf = the specific enthalpy of saturated watersfg = change in entropy during evaporationThe entropy of wet steam can be calculated as
 When steam is dry saturated, then x = 1
When steam is dry saturated, then x = 1
- Entropy of superheated steam: Consider 1 kg of dry saturated steam at Ts temperature of formation, to be superheated to a temperature Tsup.Let cps = specific heat of steam at constant pressure during superheatingChange of entropy per kg of steam during superheating
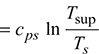 Total entropy of superheated steamssup = Entropy of dry saturated steam + Change of entropy during superheating
Total entropy of superheated steamssup = Entropy of dry saturated steam + Change of entropy during superheating
PROPERTIES OF STEAM
by
Tags:
Leave a Reply