1 Evaporation Rate
The quantity of water evaporated into steam per hour is called the evaporation rate. It is expressed as kg of steam/h, kg of steam/h/m2 of heating surface, kg of steam/h/m3 of furnace volume, or kg of steam/kg of fuel fired.
Equivalent Evaporation
It is the equivalent of the evaporation of 1 kg of water at 100°C to steam at 100°C. It requires 2257 kJ.
Factor of Evaporation
The ratio of actual heat absorption above feed-water temperature for transformation to steam (wet, dry, or superheated) to the latent heat of steam at atmospheric pressure (1.01325 bar) is known as a factor of evaporation.
Let h = specific enthalpy of steam actually produced
hf = specific enthalpy of feed water
hf (atm) = specific enthalpy of evaporation at standard atmospheric pressure
ms = actual evaporation expressed in kg/kg of fuel or kg/h of steam
me = equivalent evaporation expressed in kg/kg of fuel or kg/h
Then, the equivalent evaporation = actual evaporation × factor of evaporation
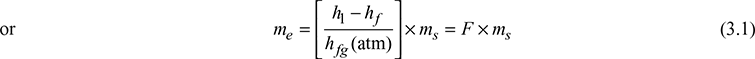
where F = factor of evaporation
h1 = hf1 + xhfg1 for wet steam actually generated
= hg1 for saturated steam actually generated
= hsup for superheated steam actually generated.
2 Performance
The performance of boiler may be explained on the basis of any of the following terms:
- Efficiency: It may be expressed as the ratio of heat output to heat input.
- Combustion rate: It is the rate of burning of fuel in kg/m3 of grate area/h.
- Combustion space: It is the furnace volume in m3/kg of fuel fired/h.
- Heat absorption: It is the equivalent evaporation from and at 100°C in kg of steam generated/m2 of heating surface.
- Heat liberated: It is the heat liberated/m3 of furnace volume/h.
3 Boiler Thermal Efficiency
It is the ratio of heat absorbed by steam from the boiler per unit time to the heat liberated by the combustion of fuel in the furnace during the same time.
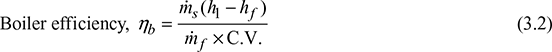
where ṁs = mass of steam generated in kg/h
ṁf = mass of fuel burned in kg/h
C.V. = calorific value of fuel in kJ/kg
4 Heat Losses in a Boiler Plant
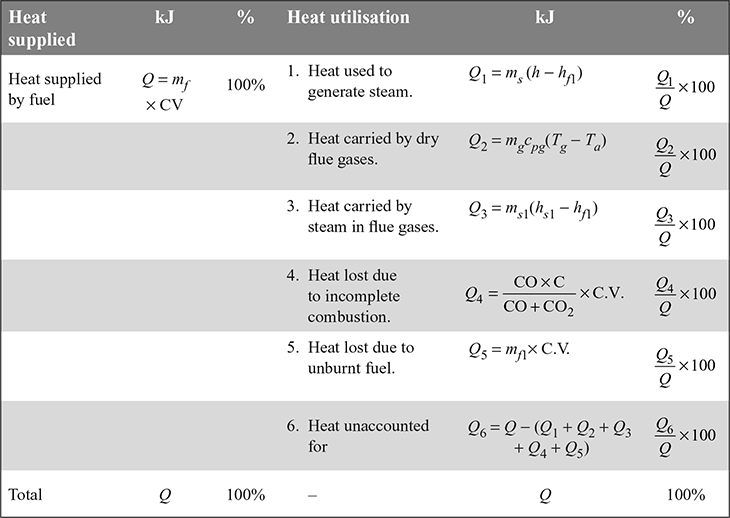
- Heat used to generate steamQ1 = ms (h − hf1)
- Heat lost to flue gases: The flue gases contain dry products of combustion and the steam generated due to the combustion of hydrogen in the fuel.Heat lost to dry flue gases,
 where mg = mass of gases formed per kg of fuelcpg = specific heat of gasesTg = temperature of gases, °CTa = temperature of air entering the combustion chamber of the boiler, °C
where mg = mass of gases formed per kg of fuelcpg = specific heat of gasesTg = temperature of gases, °CTa = temperature of air entering the combustion chamber of the boiler, °C - Heat carried by steam in flue gases
 where ms1 = mass of steam formed per kg of fuel due to combustion of H2 in fuelhf1 = enthalpy of water at boiler house temperaturehs1 = enthalpy of steam at the gas temperature and at the partial pressure of the vapour in the flue gas
where ms1 = mass of steam formed per kg of fuel due to combustion of H2 in fuelhf1 = enthalpy of water at boiler house temperaturehs1 = enthalpy of steam at the gas temperature and at the partial pressure of the vapour in the flue gas - Heat loss due to incomplete combustion: If carbon burns to CO instead of CO2, then it is known as incomplete combustion. 1 kg of C releases 10,200 kJ/kg of heat if it burns to CO, whereas it releases 35,000 kJ/kg if it burns to CO2. If the percentages of CO and CO2 in flue gases by volume are known, then
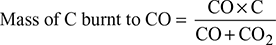 where CO, CO2 = % by volume of CO and CO2, respectively, in flue gasesC = fraction of carbon in 1 kg of fuelHeat lost due to incomplete combustion of carbon per kg of fuel,
where CO, CO2 = % by volume of CO and CO2, respectively, in flue gasesC = fraction of carbon in 1 kg of fuelHeat lost due to incomplete combustion of carbon per kg of fuel,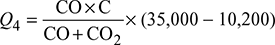
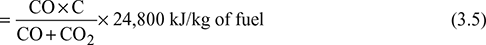
- Heat lost due to unburnt fuelQ5 = mf1 × C.V.where mf1 = mass of unburnt fuel per kg of fuel burnt.
- Convection and radiation losses: Heat unaccounted for due to convection and radiation losses,
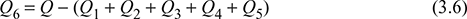 where Q = mf × C.V.= heat released per kg of fuel
where Q = mf × C.V.= heat released per kg of fuel
5 Boiler Trial and Heat Balance Sheet
There are three purposes of conducting the boiler trial.
- To determine and check the specified generating capacity of the boiler when working at full load conditions.
- To determine the thermal efficiency of the plant.
- To draw up the heat balance sheet so that suitable corrective measures may be taken to improve the efficiency.
The following measurements should be observed during the boiler trial.
- The fuel supplied and its analysis.
- Steam generated and its quality or superheat.
- Flue gases formed from exhaust analysis.
- Air inlet temperature and exhaust gases temperature.
- Volumetric analysis of exhaust gases.
- Mass of fuel left unburnt in ash.
- Feed-water temperature.
The heat balance sheet is a systematic representation of heat released from burning of fuel and heat distribution on minute, hour or per kg of fuel basis. A pro forma for heat balance sheet is given in Table 3.2.
Leave a Reply