The construction of the Orsat apparatus is shown in Fig. 1.1. It consists of three flasks a, b, and c each containing different chemicals for absorbing CO2, O2, and CO. The percentage of N2 is obtained by difference. The percentage of SO2 in the gases cannot be measured by this apparatus. The absorbents used in flask a are NaOH or KOH solution to absorb CO2. The tube b contains alkaline solution of pyrogallic acid to absorb O2. The flask c contains cuprous chloride to absorb CO. Keeping values X, Y, and Z closed, the three-way valve A is opened and the aspirator bottle B is moved down so that the flue gas enters the eudiometer tube. The flue gas is drawn in until the level in the eudiometer reads zero. The three-way valve A is closed and then the value X of the flask a is opened and the aspirator bottle is moved up and down several times pushing the gas into the flask a containing KOH solution to absorb CO2 gas. Now, the flue gas is taken into the eudiometer by lowering the aspirator bottle and the valve X is closed, keeping the original level of the solution. The aspirator is then brought near the eudiometer and placed at such a position that the water level in both places is the same and the reading of the eudiometer tube is taken. The difference in reading gives the percentage of CO2 in the flue gas. The procedure is repeated with the flasks b and c to find the percentage of O2 and CO. The remainder of the flue gas is taken as N2.
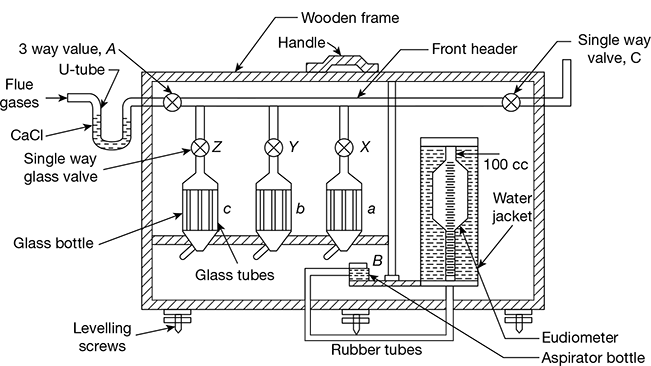
Figure 1.1 Orsat apparatus
This apparatus gives the percentages of dry flue gases only, as water vapour is condensed at room temperature when the gas is drawn over water. The flue gases may be passed through a U-tube containing calcium chloride before passing into the Orsat apparatus to ensure that only dry gas is being analysed.
Leave a Reply