The cooling of flue gases to room temperature is not possible in most combustion processes. Therefore, the amount of latent heat of water vapour goes waste. Accordingly, for the calculation of the lower calorific value, we assume that the water vapour formed during combustion leaves as vapour.
LCV = HCV − heat carried away by water vapour formed per kg of fuel burned.

where mw = mass of water vapour formed per kg fuel burned, hfg = latent heat of vapourisation at the partial pressure of water vapour in the combustion products = 2395 kJ/kg.
The HCV of fuel is given by
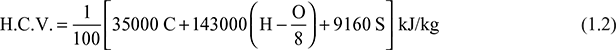
where C, H, O, and S are percentages of carbon, hydrogen, oxygen, and sulphur, respectively.
The LCV of fuel is given by
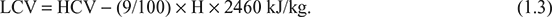
Leave a Reply