The temperature-entropy diagram is shown in Fig. 20.2.
Enthalpy of moist air = Enthalpy of dry air + Enthalpy of water vapour associated with dry air
h = ha + whv per kg of dry air
where ha = cpa td = 1.005 td kJ/kg
cpa = 1.005 kJ/kg. K
td = DBT of air in °C.
hv = cpw tdp +[(hfg)dp + cpv (td −tdp)]kJ/kg
= 4.1868tdp + (hfg)dp +1.88(td −tdp)
where cpw = 4.1868 kJ/kg K is the specific heat of liquid water
(hfg)dp = latent heat of vaporization at DPT
cpv = 1.88 kJ/kg.K is the specific heat of superheated water vapour
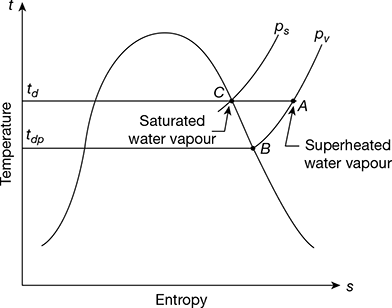
Figure 20.2 Temperature-entropy diagram
tdp = DPT, °C
w = specific humidity
h = 1.005 td + w [4.1868 tdp + (hfg)dp + 1.88 (td + tdp)]
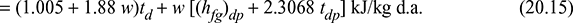
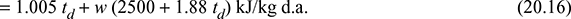
Leave a Reply