Heat Engine: In a heat engine, as shown in Fig. 18.4(a), the heat supplied to the engine is converted into useful work. If Q2 is the heat supplied to the engine and Q1 is the heat rejected from the engine, then the net work done by the engine is given by,
We =Q2 −Q1
Efficiency of heat engine,
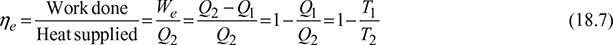
Refrigerator: A refrigerator is an equipment used to remove the heat continuously from the space (sink). It maintains the temperature below atmospheric temperature and reject the heat to the atmosphere (source) by increasing the temperature potential of the heat to be rejected with the help of mechanical work input (compressor).
The refrigeration system is shown in Fig. 18.4(b). In a refrigerator system Q1 quantity of heat is removed from the source where its temperature T1 is maintained below atmospheric temperature T2. COP of refrigerator is,
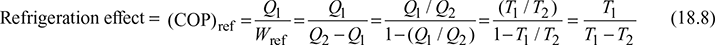
Heat Pump: It is an equipment used to supply the heat continuously to the space. It maintains the temperature above atmospheric temperature by absorbing the heat from the atmosphere and increasing its temperature potential with mechanical work input (compressor). The heat pump system is shown in Fig. 18.4(c). In a heat pump system, Q2 quantity of heat is supplied to the room where the temperature T2 is maintained above atmospheric temperature T1. The heat Q1 is obtained from the atmosphere.
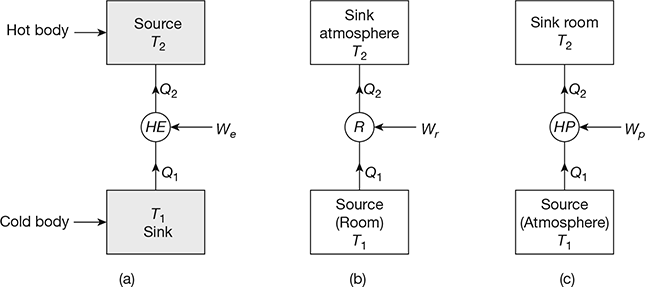
Figure 18.4 (a) Heat engine, T1> T>atm, (b) Refridgerator T1 < Tatm, (c) Heat pump
COP or Energy performance ratio of heat pump is,
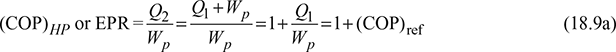
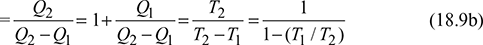
Example 18.1
Find the COP of a refrigeration system if the work input is 10 kJ/kg and refrigeration effect produced is 180 kJ/kg of refrigerant flowing.
Solution
wref =100 kJ/kg, q=180 kJ/kg
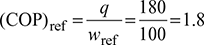
Example 18.2
A machine working on a Carnot cycle operates between 310 K and 270 K. Calculate the COP when it is operated as:
- a refrigerator, and
- a heat pump.
Solution
Given: T1 = 270 K, T2 = 310 K
- Refrigerator
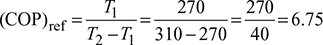
- Heat pump:
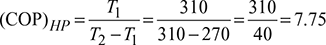
Example 18.3
A Carnot refrigeration cycle absorbs heat at 268 K and rejects it at 298 K.
- Calculate the COP of this refrigeration cycle.
- If the cycle is absorbing 1150 kJ/min at 268 K, find the rate of work input required.
- How many kJ/s will be delivered if it works as a heat pump at 298 K and absorbs 1150 kJ/min at 268 K.
Solution
Given: T1 = 268 K, T2 = 298 K, Q1 = 1150 kJ/min
- Carnot refrigeration cycle:
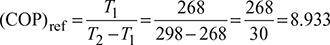 Work required,
Work required, 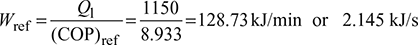
- Heat pump:(COP)HP =1 + (COP)ref =1 + 8.933= 9.933
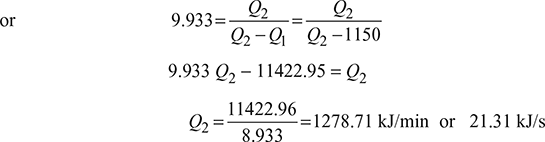
Leave a Reply