Let the volumetric analysis of gaseous fuel be known. The chemical equations for gaseous fuel are as follows:
- 2H2 + O2 → 2H2O2 volumes of H2 + 1 volume of O2 → 2 volumes of H2Oor 1 volume of H2 +
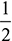 volume of O2 → 1 volume of H2O
volume of O2 → 1 volume of H2O - 2CO + O2 → 2CO22 volumes of CO + 1 volume of O2 → 2 volumes of CO2
- CH4 + 2O2 → 2H2O + CO21 volume of CH4 + 2 volumes of O2 → 2 volumes of H2O + 1 volume of CO2
- C2H4 + 3O2 → 2H2O + 2CO21 volume of C2H4 + 3 volumes of O2 → 2 volumes of H2O + 2 volumes of CO2
Let the volume fraction of the gaseous fuel contain H2, CO, CH4, C2H4, CO2, and N2, the last two being incombustible gases.
The quantity of O2 required ![]() m3/m3 of gas
m3/m3 of gas
Air contains 21% of O2 by volume.
Quantity of air required ![]()
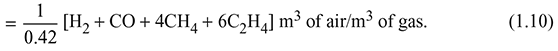
Leave a Reply