1 Percentage of Carbon by Mass in Fuel and Volumetric Analysis is Known
We know that 1 kg of flue gas contains
![]() kg of CO and
kg of CO and ![]() kg of CO2
kg of CO2
Amount of carbon in ![]() kg of
kg of ![]()
Amount of carbon in ![]() kg of
kg of ![]()
Total mass of carbon in 1 kg of flue gas ![]() kg
kg
Let C = mass fraction of carbon in fuel and there is no nitrogen in fuel.
mg = mass of flue gases formed per kg of fuel burned.
Then, ![]()

Now, nitrogen in 1 kg of air supplied = 0.77 kg
N2 in 1 kg of flue gas = ![]()
∴ Air supplied per kg of flue gas = ![]()
Mass of air supplied per kg of fuel = mass of air supplied per kg of flue gas × mass of flue gases formed per kg of fuel.
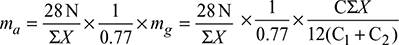

2 Excess Air Supplied
The oxygen carried in the flue gases comes only from the air supplied for the combustion of fuel. In addition, the oxygen necessary to convert CO to CO2 should be taken into account in calculating the excess air supplied.
CO in 1 kg of flue gas = ![]()
O2 in 1 kg of flue gas = ![]()
O2 required for burning ![]() kg of CO to CO2 =
kg of CO to CO2 = ![]()
Excess O2 available per kg of flue gas formed ![]()
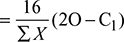
Excess O2 supplied per kg of fuel = Excess O2 per kg of flue gas × mg
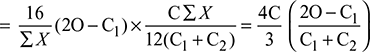
Excess air supplied per kg of fuel
Leave a Reply