Dalton’s law states that
- The total pressure in a container having mixture of gas and vapour is the sum of the partial pressure of the vapour at the common temperature and the partial pressure of the gas.If the degree of saturation of air at any point inside the condenser is 100%, the partial pressure of vapour is the same as the saturation pressure.Let t°C = temperature of mixture in a container.pa = absolute pressure of any gas or air at t°C.ps = saturation pressure of any vapour corresponding to t°C.pt = total pressure in the container
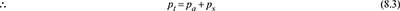
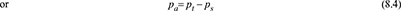
- Each constituent of the mixture in the container occupies the whole volume of the container and exerts its own partial pressure.Let V = volume of a container, m3ma = mass of air in the container, kgmg = mass of water vapour in the container, kgm = total mass of mixture in the container, kgvg = specific volume of saturated vapour at t°C and pa in m3/kgva = specific volume of air at t°C in m3/kg
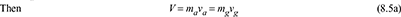
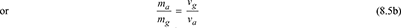

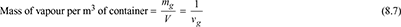
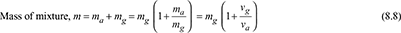

Example 8.1
A vacuum of 67 cm of Hg was obtained with the barometer reading of 75 cm of Hg. The condensate temperature is 20°C. Correct the vacuum to a standard barometer of 76 cm and hence determine the partial pressure of air and steam. Also find the mass of air present with 1 kg of steam.
Solution
Corrected vacuum = 76 − (actual barometric reading − actual vacuum)
= 76 − (75 − 67) = 68 cm of Hg
Pressure of dry saturated steam at 20°C from steam tables is 0.023385 bar
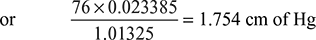
and specific volume is v = 57.79 m3/kg
Total absolute pressure in the condenser = 75 − 67 = 8 cm of Hg
Partial pressure of steam, pg = 1.754 cm of Hg
∴ Partial pressure of air, pa = 8 − 1.754 = 6.246 cm of Hg
According to Dalton’s law the volume of air present per kg of steam
= 57.79 m3/kg at 20°C
Also for air paV = maRT
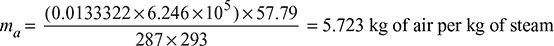
Leave a Reply