There is a need to recycle the cooling water through the condenser when cooling water supply is limited. Cooling tower is an artificial device used to cool hot cooling water coming out from the condenser.
There are different types of cooling towers. The induced draft type cooling towers are commonly used in high capacity power plants. The schematic diagram of an induced draft cooling tower is shown in Fig. 8.12. The hot water coming out from the condenser is sprayed at the top of the tower and air is induced to flow through the tower with the help of induced draft fans mounted at the top of the tower.
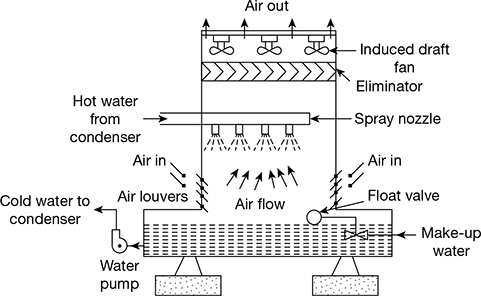
Figure 8.12 duced draft cooling tower
The amount of water supply lost in such a tower ranges from 1% − 2% by evaporation and 0.5% − 2% by drift losses. To compensate these losses, make-up water is supplied from external sources. The cooled water is collected at the bottom.
Example 8.3
The following readings were taken during a test on a surface condenser:
Vacuum in condenser = 71 cm of Hg
Barometer reading = 76 cm of Hg
Temperature in condenser = 33°C
Hot well temperature = 30°C
Cooling water circulated = 48,000 kg/h
Inlet temperature of cooling water = 15°C
Outlet temperature of cooling water = 28°C
Condensate = 1500 kg/h
Calculate:
- the mass of air in kg/m3 of condensate volume,
- dryness fraction of steam entering the condenser, and
- the vacuum efficiency
Solution
- Absolute pressure in condenser,pt = (76 − 71) × 0.01333 = 0.0667 barAt t = 33°C, ps = 0.05106 bar from steam tables = 3.83 cm of HgPartial pressure of air, pa = pt − ps= 0.0667 − 0.05106 = 0.01564 bar
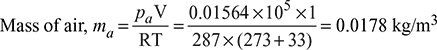
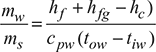 At p = 0.0667 bar, hf = 159.47 kJ/kg, hfg = 2411.4 kJ/kg
At p = 0.0667 bar, hf = 159.47 kJ/kg, hfg = 2411.4 kJ/kg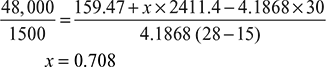
- Vacuum efficiency
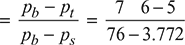 = 0.9837 or 98.37%
= 0.9837 or 98.37%
Example 8.4
A steam turbine discharges 6000 kg of steam per hour at 45°C and 0.82 dryness. The estimated air leakage is 15 kg/h. The temperature at the suction of air pump is 30°C and temperature of condensate is 33°C. Find (a) the vacuum gauge reading, (b) capacity of air pump, (c) loss of condensate per hour, and (d) the quantity of cooling water required by limiting its temperature rise by 10°C.
Solution
- Total pressure in the condenser, pt = pa + psAt 45°C, ps = 0.095934 bar from steam tables vg = 15.258 m3/kgVolume of 6000 kg steam, V = 6000 ×xvg = 6000 × 0.82 × 15.258 = 75069.36 m3/h= volume of airPartial pressure of air,
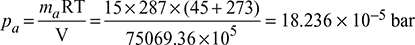 pt = 18.236× 10-5 + 0.09582 = 0.096 barVacuum in condenser
pt = 18.236× 10-5 + 0.09582 = 0.096 barVacuum in condenser 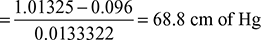
- Partial pressure of steam at air pump suction at 30°C = 0.04242 barpa = pt − ps = 0.096 − 0.042461 = 0.0535 barVolume of air at 30°C and 0.0535 bar,
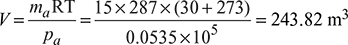 Air pump capacity = 243.82 m3/h
Air pump capacity = 243.82 m3/h - Specific volume of steam at 30°C, vg = 32.893 m3/kg
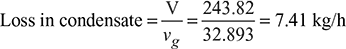
- At 45°C, hf = 188.42 kJ/kg, hfg = 2394.8 kJ/kgMass of cooling water,
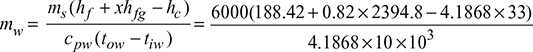 = 288.62 tonnes/h
= 288.62 tonnes/h
Example 8.5
The air leakage into a surface condenser operating with a steam turbine is estimated as 80 kg/h. The vacuum near the inlet of air pump is 70 cm of Hg when barometer reads 76 cm of Hg. The temperature at the inlet of vacuum pump is 20°C. Calculate (a) the minimum capacity of the air pump, (b) dimensions of the reciprocating air pump to remove air if it runs at 200 rpm. Take L:D:: 3:2 and volume efficiency = 98%, (c) mass of vapours extracted per hour.
Solution
- Total pressure at inlet of air pump at 1–1, (Fig. 8.13)
 Partial pressure of water vapour at 1–1 corresponding to saturation temperature of 20°C, from steam tables,ps = 0.023385 barPartial pressure of air at 1–1,pa = pt − ps = 0.08 − 0.023385 = 0.0566 barVolume of 80 kg/h of air at 20°C and 0.0566 bar,
Partial pressure of water vapour at 1–1 corresponding to saturation temperature of 20°C, from steam tables,ps = 0.023385 barPartial pressure of air at 1–1,pa = pt − ps = 0.08 − 0.023385 = 0.0566 barVolume of 80 kg/h of air at 20°C and 0.0566 bar,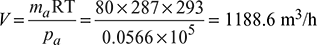 Capacity of air pump = 1188.6 m3/h
Capacity of air pump = 1188.6 m3/h - Now V =
 D2LNηv
D2LNηv 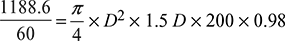 D3 = 0.0858D = 0.441 m or 46.1 cmL = 66.2 cm
D3 = 0.0858D = 0.441 m or 46.1 cmL = 66.2 cm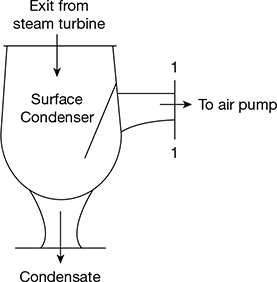 Figure 8.13 Surface condenser
Figure 8.13 Surface condenser - Mass of water vapour going with air in the air pump,
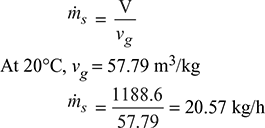
Example 8.6
The quality of steam entering a jet condenser is 0.92. It is condensed by using cooling water at 15°C. The mass of air in the condenser is 35% of the mixture. Assuming that only latent heat of steam is absorbed by the cooling water, calculate (a) the temperature of mixture of condensate and water leaving the condenser, and (b) the mass of water required per kg of steam condensed.
Take for air, R = 287 J/kg.K and for steam, R = 463 J/kg.K. Condenser vacuum = 61 cm of Hg, Barometer reading = 76 cm of Hg.
Solution
- Given that pa = 0.35 ptpt = (76 − 61) × 0.0133 = 0.2 barpa = 0.35 × 0.2 = 0.07 barps = pt − pa = 0.2 − 0.07 = 0.13 barSaturation temperature of steam at 0.13 bar, from steam tables,ts = 50.71°CAs only latent heat of steam is to be absorbed, therefore, outlet temperature of cooling water,tow = ts = 50.71°CSince steam and cooling water mix together in the jet condenser,Mixture temperature coming out of condenser = 50.71°C
- Now
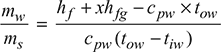 At ps = 0.13 bar, hf = 212.27 kJ/kg, hfg = 2380.98 kJ/kg from steam tables
At ps = 0.13 bar, hf = 212.27 kJ/kg, hfg = 2380.98 kJ/kg from steam tables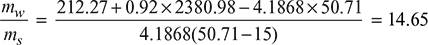 Minimum quantity of cooling water required per kg of steam condensed= 14.65 kg
Minimum quantity of cooling water required per kg of steam condensed= 14.65 kg
Example 8.7
In a trial on a surface condenser, the vacuum gauge reading was 660 mm of Hg when barometer read 760 mm of Hg. The temperature in the condenser was 45°C.
- Determine the alteration in vacuum if the quantity of air entering the condenser is reduced by 100 kg/h.
- If the mass of condensate is 960 kg/h, calculate the quantities of air and vapour which the pump has to handle.
Solution
Absolute condenser pressure = 760 − 660 = 100 mm of Hg
pt = ![]() × 1.01325 = 0.1333 bar
× 1.01325 = 0.1333 bar
At 45°C, from steam tables,
Partial pressure of steam, ps = 0.095934 bar
Specific volume of steam, vsg = 15.258 m3/kg
Partial pressure of air, pa = 0.1333 − 0.095934 = 0.037366 bar
Volume of steam per hour, Vs = 960 × 15.258 = 14647.68 m3
= Volume of air, Va
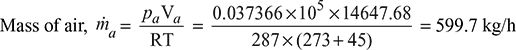
If the mass of air is reduced to 100 kg/h, then partial pressure of air,
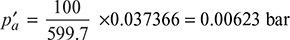
New absolute pressure of condenser, p′t = 0.095934 + 0.00623 = 0.102 bar
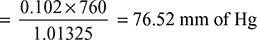
New vacuum reading = 760 − 76.52 = 683.48 mm Hg
Alteration in vacuum = 683.48 − 660 = 23.48 mm Hg
Capacity of pump = 960 + 100 = 1060 kg/h
Example 8.8
The following observations were recorded during a condenser test;
Vacuum reading = 700 mm of Hg; Barometer reading = 760 mm of Hg;
Condensate temperature = 34°C
Find: (a) partial pressure of air and (b) mass of air per m3 of condenser volume.
Take R for air = 287 J/kg K
Solution
Given that Vacuum reading = 700 mm of Hg: Barometer reading = 760 mm of Hg; T = 34°C = 34 + 273 = 307 K
- Pressure in the condenserpc = Barometer reading − Vacuum reading= 760 − 700 = 60 mm of Hg= 60 × 0.00133 = 0.0798 bar (∵ 1 mm of Hg = 0.001 33 bar)From steam tables, corresponding to a temperature of 34°C, we find that pressure of steam,ps = 0.0535 bar∴ Partial pressure of air,pa = pc − ps = 0.0798 − 0.0535 = 0.0263 bar
- Mass of air per m3 of condenser volume.
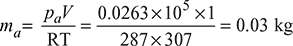
Example 8.9
A vacuum gauge fitted to a condenser reads 680 mm of Hg, when the barometer reads 750 mm of Hg. Determine the corrected vacuum in terms of mm of Hg and bar.
Solution
Given that Vacuum gauge reading = 680 mm of Hg; Barometer reading = 750 mm of Hg:
Pressure in the condenser = 750 − 680 = 70 mm of Hg
Corrected vacuum = 760 − 70 = 690 mm of Hg
= 690 × 0.00133 = 0.918 bar
Example 8.10
Calculate the vacuum efficiency from the following data:
Vacuum at steam inlet to condenser = 700 mm of Hg
Barometer reading = 760 mm of Hg
Hot well temperature = 30°C
Solution
Given that Vacuum reading or actual vacuum = 700 mm of Hg; Barometer reading = 760 mm of Hg; t = 30°C
Pressure in the condenser = 760 − 700 = 60 mm of Hg
From steam tables, corresponding to a temperature of 30°C, we find that ideal pressure of steam,
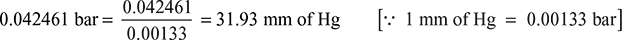
Ideal vacuum = Barometer reading − Ideal pressure
= 760 − 31.93 = 728.07 mm of Hg
Vacuum efficiency, ηvac = ![]() = 0.9614 or 96.14%
= 0.9614 or 96.14%
Example 8.11
The vacuum efficiency of a condenser is 96%. The temperature of condensate is 40°C. If the barometer reads 752 mm of Hg, find the vacuum gauge reading of the condenser.
Solution
Given that ηv = 96% = 0.96%; t = 40°C;
We find that ideal pressure of steam, = 0.073837 bar = ![]() = 55.52 mm of Hg
= 55.52 mm of Hg
∴ Ideal vacuum = Barometer reading − Ideal pressure
= 752 − 55.52 = 696.48 mm of Hg

∴ Actual vacuum or vacuum gauge reading of the condenser
= 0.96 × 696.48 = 668.62 mm of Hg
Example 8.12
In a surface condenser, the vacuum maintained is 700 mm of Hg. The barometer reads 754 mm. If the temperature of condensate is 18°C, determine: (a) mass of air per kg of steam and (b) vacuum efficiency.
Solution
Given that Actual vacuum = 700 mm of Hg; Barometer reading = 754 mm of Hg; T = 18°C = 18 + 273 = 291 K
Pressure in the condenser, pc = 754 − 700 = 54 mm of Hg
From steam tables, corresponding to 18°C, we find that absolute or ideal pressure of steam,
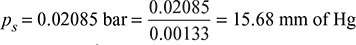
Specific volume of steam, vs = 65.844 m3/kg
- Pressure of air (as per Dalton’s law)pa = pc − ps = 54 − 15.68 = 38.32 mm of Hg= 38.32 × 0.00133 = 0.051 bar = 0.051 × 105 N/m2Mass of air per kg of steam,
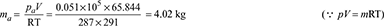
- Ideal vacuum = Barometer reading − Ideal pressure= 754 − 15.68 = 738.32 mm of Hg

Example 8.13
A surface condenser, fitted with separate air and water extraction pumps, has a portion of the tubes near the air pump suction screened off from the steam so that the air is cooled below the condensate temperature. The steam enters the condenser at 38°C and the condensate is removed at 37°C. The air removed has a temperature of 36°C. (a) If the total air infiltration from all sources together is 5 kg/h, determine the volume of air handled by the air pump per hour. (b) What would be the corresponding value of the air handled if a combined air and condensate pump was employed? Assume uniform pressure in the condenser.
Solution
Given that Ts = 38°C = 38 + 273 = 311 K; Tc = 37°C = 37 + 273 = 310 K; Ta = 36°C = 36 + 273 = 309 K; ṁa = 5 kg/h
- Since the pressure at entry to the condenser (pc) is equal to the pressure of steam corresponding to 38°C, therefore, from the steam tables,pc = 0.0668 barPressure of steam at the air pump suction, corresponding to 36°C (from steam tables),ps = 0.0598 bar∴ Pressure of air at the air pump suction (as per Dalton’s law),pa = pc − ps = 0.0668 − 0.0598 = 0.007 bar= 0.007 × 105 = 700 N/m2Volume of air handled by the air pump,
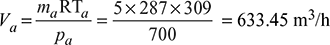
- From steam tables corresponding to a condensate temperature of 37°C, we find that pressure of steam,ps = 0.0633 bar∴ Pressure of air (as per Dalton’s law),pa = pc − ps = 0.0668 − 0.0633 = 0.0035 bar= 0.0035 × 105 = 350 N/m2Volume of air handled,
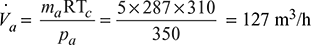
Example 8.14
The air leakage into a surface condenser operating with a steam turbine is estimated as 84 kg/h. The vacuum near the inlet of air pump is 700 mm of Hg when barometer reads 760 mm of Hg. The temperature at inlet of vacuum pump is 20°C. Calculate the following:
- The minimum capacity of the air pump in m3/h;
- The dimensions of reciprocating air pump to remove the air if it runs at 200 rpm. Take L/D ratio = 1.5 and volumetric efficiency = 100%
- The mass of vapour extracted per minute
Solution
Given that ṁa = 84 kg/h; Vacuum = 700 mm of Hg; Barometer reading = 760 mm of Hg; T = 20°C = 20 + 273 = 293 K
- Pressure in the condenser,pc = Barometer reading − Condenser vacuum = 760 − 700 = 60 mm of Hg = 60 × 0.00133 = 0.0798 barFrom steam tables, corresponding to a temperature of 20°C, we find that pressure of steam,ps = 0.023385 bar∴ Pressure of air (as per Dalton’s law),pa = pc − ps = 0.0798 − 0.023385 = 0.056415 bar= 0.056415 × 105 = 5641.5 N/m2Minimum capacity of the air pump,
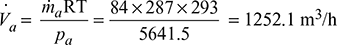
- Let D = Diameter of the cylinder in metre,L = Length of the stroke in metres = 1.5 Dηv = Volumetric efficiency = 100% = 1N = Speed of the pump = 200 r.p.m.Minimum capacity of the air pump (Va),
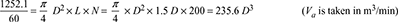 ∵ D3 = 0.0886 or D = 0.446 mand L = 1.5 D = 1.5 × 0.446 = 0.669 m
∵ D3 = 0.0886 or D = 0.446 mand L = 1.5 D = 1.5 × 0.446 = 0.669 m - From steam tables, corresponding to a temperature of 20°C, we find that specific volume of steam,vg = 57.79 m3/kg∴ Mass of vapour extracted per minute
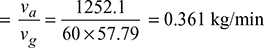
Example 8.15
The vacuum at the extraction pipe in a condenser is 710 mm of mercury and the temperature is 35°C. The barometer reads 760 mm of mercury. The air leakage into the condenser is 4 kg per 10,000 kg of steam. Determine: (a) the volume of air to be dealt with by the dry air pump per kg of steam entering the condenser, and (b) the mass of water vapour associated with this air. Take R = 287 J/kg K for air.
Solution
Given that vacuum = 710 mm of Hg; T = 35°C = 35 + 273 = 308 K; Barometer reading = 760 mm of Hg; ma = 4 kg per 10000 kg of steam = 0.0004 kg/kg of steam.
- Let Va = Volume of air per kg of steam entering the condenser.Pressure in the condenserpc = Barometer reading − Condenser vacuum= 760 − 710 = 50 mm of Hg = 50 × 0.001 33= 0.0065 barFrom steam tables, corresponding to the temperature of 35°C, we find that the pressure of steam,ps = 0.05628 bar∴ Pressure of air (as per Dalton’s law),pa = pc − ps = 0.0665 − 0.05628 = 0.01022 bar= 0.01022 × 105 = 1022 N/m2Now paVa = ma RT
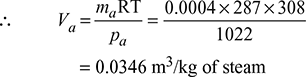
- From steam tables, corresponding to a temperature of 35°C, we find that specific volume of steam,vg = 25.216 m3/kg∴ Mass of water vapour associated with the air
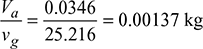
Example 8.16
The amount of steam condensed in a condenser is 6800 kg/h and the air leakage into the condenser is 12 kg/h. The air pump suction is screened off. The exhaust steam temperature is 32°C, the condensate temperature is 30°C and the temperature at the air pump suction is 25°C. Determine the following:
- The mass condensed in the air cooler section per hour
- The volume of air in m3 handled by air pump
- The percentage reduction in air pump capacity due to cooling of the air.
Solution
The saturation pressure corresponding to steam temperature 32°C from steam table is
pa = 0.04799 bar
Specific volume corresponding to 32°C, vs = 29.82 m3/kg
∴ Volume of steam handled, Vs = 6800 × 29.82 = 202776 m3/h
According to Dalton’s law, the volume of air handled per hour is equal to the volume of steam handled per hour. Hence the partial pressure of air,
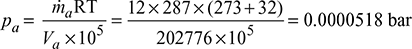
We observe that the value of partial pressure of air is very small and hence the total condenser pressure may be taken as due to steam alone i.e. 0.04799 bar.
Since the temperature at the air pump suction is 25°C, hence the steam pressure at the air pump suction corresponding to 25°C from steam tables,
ps = 0.031691 bar
Specific volume corresponding to 25°C, vs = 43.359 m3/kg
∴ Partial pressure of air handled,
pa = 0.04799 – 0.031691 = 0.0163 bar
∴ Volume of air handled per hour by the pump,
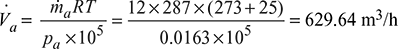
According to Dalton’s law, the volume of steam will also be equal to 629.64 m3/h.
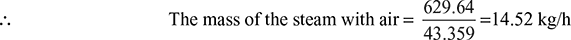
The temperature may be assumed to be that of the condensate, i.e., ts = 30°C at the point where the saturated steam enters the air cooling section. Corresponding to 30°C, the pressure is:
ps = 0.042461 bar
Specific volume, vs = 32.893 m3/h
∴ Partial pressure of air is given by,
pa = (0.04799 − 0.042461) = 0.00553 bar
Hence, the volume of air handled
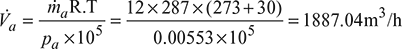
According to Dalton’s law, this volume of air is equal to the volume of steam hence the mass of steam associated with the air,
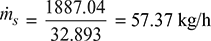
∴ Mass of steam condensed in the cooler section
= 57.37 − 14.52 = 42.85 kg/h
Hence, the percentage reduction in volume handled by the air pump due to cooling section is:
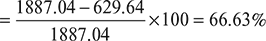
Example 8.17
In a condenser test, the following observations were made
Vacuum = 70 cm of Hg
Barometer = 76 cm of Hg
Mean temperature of condensation = 35°C
Hot well temperature = 29°C
Mass of cooling water = 45500 kg/h
Inlet water temperature = 16.5°C
Outlet water temperature = 31°C
Mass of condensate = 1200 kg/h
Find (a) the mass of air present per unit condenser volume, (b) the state of steam entering the condenser, (c) the vacuum efficiency, (d) the condensate undercooling and (e) condenser efficiency.
Solution
- Corresponding to 35°C, the partial pressure of steam from steam table = 0.05628 barAbsolute pressure of mixture in condenser = (76 − 70) ×
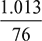 = 0.07997 bar∴ Partial pressure of air = 0.07997 − 0.05628 = 0.02369 barMass of air =
= 0.07997 bar∴ Partial pressure of air = 0.07997 − 0.05628 = 0.02369 barMass of air = 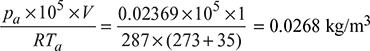
- Heat absorbed by circulating water = 45500 × 4.18 (31 − 16.5) = 2727755 kJ/hHeat rejected by steam in condensing at 35°C and in being under cooled from 35°C to 29°C = mc (hw1 + xL1 − hwc) = 1200 (146.66 + 2418.6x − 121.59) = 1200 (25.07 + 2418.6x)Now, Heat absorbed by circulating water = Heat rejected by steam
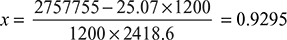
- Vacuum efficiency
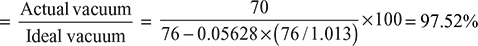
- Condensate undercooling = 35 − 29 = 6°C
- Condenser efficiency =

Example 8.18
In a surface condenser 2500 kg/h of steam is condensed and the air leakage is 2 kg/h. A separate air and water extraction pumps are fitted which draws the air in air cooler and a portion of the tube near the air pump suction is screened off from the steam so that the air is cooled below the condensate temperature. The steam enters the condenser dry and saturated at 38°C and the condensate is extracted at the lowest point of the condenser at a temperature of 37°C (i.e., the temperature at the entrance of air cooler is 3°C). The temperature at the air pump suction is 31°C. Assuming a constant vacuum throughout the condenser, find the following:
- The mass of steam condensed/min in the air cooler
- The volume of air to be dealt with per minute by the dry pump
- The reduction in the necessary air pump capacity following the cooling of air.
Solution
Partial pressure of steam at 38°C, ps = 0.0668 bar
Specific volume of steam at 38°C, vs = 21.8 m3/kg
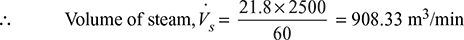
This must be the volume of 2 kg/h of air when exerting its partial pressure, i.e., Va = Vs = 908.33 m3/min
Partial pressure of air, pa =![]() = 0.000033 bar
= 0.000033 bar
Hence, total pressure in the condenser = ps + pa = 0.0668 + 0.000033 = 0.06683 bar
At condensate extraction:
Partial pressure of steam at 37°C = 0.0633 bar
Specific volume of steam at 37°C, vs = 22.94 m3/kg
∴ Partial pressure of air = 0.06683 − 0.0633 bar = 0.00353 bar
∴ Volume of air, V̇a =![]() = 8.473 m3/min = Vs
= 8.473 m3/min = Vs
∴ The mass of the steam associated with air,
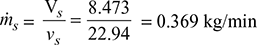
At air pump suction:
Partial pressure of steam of 31°C = 0.04522 bar
∴ Partial pressure of air = 0.06683 – 0.04522 = 0.02161 bar
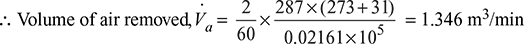 Mass of steam associated with this air and which is partially condensed in the air cooler
Mass of steam associated with this air and which is partially condensed in the air cooler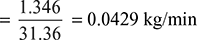
- Mass of steam condensed in the air cooler or saving in condensate by using the separate extraction method= 0.369 − 0.0429 = 0.3261 kg/min
- Air capacity without air cooler = 8.473 m3/minAir pump capacity with air cooler = 1.346 m3/min∴ Percentage reduction in pump capacity
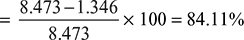
Example 8.19
A barometric jet condenser maintains a vacuum of 64.2 cm of Hg when the barometer reads 76 cm of Hg. The condenser handles 4600 kg of steam/h and 0.97 dryness fraction. The inlet temperature of the cooling water is 15°C. The mixture of condensate and cooling water leaves at 43°C. Calculate (a) the minimum height of the tail pipe above the level of the hot well and (b) the amount of cooling water required. Assume no undercooling.
Solution
- Absolute condenser pressure of steam = (76 − 64.2) × 0.0133 = 1.6048 m of waterBarometer height = 76 ×
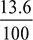 = 10.336 m of waterHence the length of the tail pipe = 10.336 − 1.6048 = 8.7312 m
= 10.336 m of waterHence the length of the tail pipe = 10.336 − 1.6048 = 8.7312 m - The saturation temperature of steam entering the condenser will be 43°C in the absence of any undercooling of the condensate. Corresponding to 43°C,
Example 8.20
A condenser is equipped in a steam turbine which handles 14500 kg of steam per hour and develops 2484.3 kW. The initial conditions of steam entering to turbine are 27 bar and 350°C. The exhaust from the turbine is condensed in the condenser and the vacuum maintained is 72.5 cm of Hg while barometer reads 75.8 cm of Hg. The temperature of the circulating water is increased from 8 to 28°C while the condensate is removed from the condenser at a temperature of 29°C. Calculate the following:
- Dryness fraction of steam entering the condenser
- Mass of circulating water per hour and the cooling ratio
- Minimum quantity of cooling water required per kg of steam.
Solution
- Absolute condenser pressure = (75.8 − 72.5) × 1.013/76 = 0.04398 barFrom the steam tables, corresponding to 0.043398 bar, hf = 327.19 kJ/kg and hfg = 2313.46 kJ/kgCorresponding to 27 bar and 350°C, h = 3124.84 kJ/kgIf x be the dryness fraction of steam entering the condenser, then drop of enthalpy
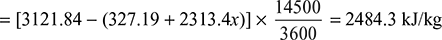 ∴ x = 0.9414
∴ x = 0.9414 - Heat given by steam = Heat received by water14500[(121.48 + 0.9414 × 2433.1) − 117.31] = mw × 4.18 (28 − 8)Mass of cooling water = mw = 398002.5 kg/h
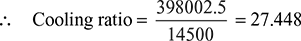
- The maximum possible temperature of cooling water at outlet will be 29.4°C corresponding to condenser pressure of 0.04398 bar.Hence maximum rise in temperature of cooling water = 29.4 − 8 = 21.4°CIf under this condition mw1 be the quantity of cooling water required per kg of steam, by the heat balance equation, then[(123.22 + 0.942 × 2432.4 − 121.48)] mw1 × 4.18 × 21.4mw1 = 25.634 kg of water/kg of steam
Example 8.21
The following observations were recorded during a trial on a steam condenser:
Condenser vacuum recorded = 70 cm (0.93325 bar)
Barometer reading = 76.5 cm Hg (1.02 bar)
Mean condenser temperature = 35°C
Hot well temperature = 28°C
Condensate formed/hr. = 1800 kg
Circulating cooling water inlet temperature = 15°C
Circulating cooling water outlet temperature = 27°C
Quantity of cooling water/h = 80000 kg
Determine the following:
- Vacuum corrected to standard barometer of 76 cm Hg (1.01325 bar)
- Vacuum efficiency
- Under cooling of condensate
- Condenser efficiency
- State of steam entering condenser
- Mass of air per cubic metre of condenser volume.
- Mass of air present per kg of uncondensed steam
Assume R for air as 287 kJ/kgK.
Solution
- Vacuum corrected to standard barometer= Standard barometric pressure − (Barometric pressure − Gauge pressure)= 1.01325 bar − (1.02 bar − 0.93335 bar)= 1.01325 − 0.8665 = 0.9266 bar
- From steam table, the pressure corresponding to condenser temperature of 35°C = 0.0562 bar
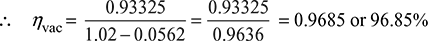
- Undercooling of condensate = 35° − 28° = 7°C
- Absolute condenser pressure = Barometer pressure − Vacuum reading= 1.02 − 0.93325 = 0.08675 barSaturation temperature corresponding to pressure of 0.08665 bar from steam tables is 42.9°CThus, cooling water temperature can get raised to 42.9°C

- From steam tables for pressure of 0.08665 barhf = 179.8 kJ/kg, hfg = 2399.6 kJ/kg.Enthalpy of condensate corresponding to hot well temperature of 28°C is 117 kJ/kg.Heat absorbed by cooling water = Heat given by steammcpw (t2 − t1) = ms (hf + xhfg − hc)
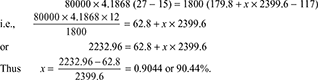
- At condenser temperature of 35°C, the partial pressure of steam,ps = 0.0562 bar.Hence, ma =
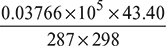 = 1.911 kg per kg of vapouror mv =
= 1.911 kg per kg of vapouror mv = 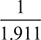 = 0.523 kg of vapour per kg of air.With external cooling from 25°C to 20°C we have, from steam tables, at 20°Cps = 0.023366 bar = 1.7526 cm Hg, vg = 57.834 m3∴ pa = 5.2 − 1.7526 = 3.4474 cm Hg
= 0.523 kg of vapour per kg of air.With external cooling from 25°C to 20°C we have, from steam tables, at 20°Cps = 0.023366 bar = 1.7526 cm Hg, vg = 57.834 m3∴ pa = 5.2 − 1.7526 = 3.4474 cm Hg 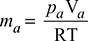 where pa = 3.4474 ×
where pa = 3.4474 × 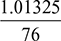 = 0.04596 barVa = vg = 57.834 m3 per kg of vapourR = 287 J/kg ×K; T = (273 + 20) = 293 KHence, ma =
= 0.04596 barVa = vg = 57.834 m3 per kg of vapourR = 287 J/kg ×K; T = (273 + 20) = 293 KHence, ma = 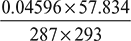 = 3.161 kg per kg of vapourmv =
= 3.161 kg per kg of vapourmv = 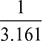 = 0.316 kg of vapour per kg of air
= 0.316 kg of vapour per kg of air
These calculations show that by cooling the mixture form 25°C to 20°C, vapour accompanying each kg of air withdrawn from the condenser is reduced from 0.523 kg to 0.316 kg. This will have a significant effect both on the ejector capacity and work done for air pump suction.
Example 8.22
In a condenser test, the following observations were made:
Vacuum = 690 mm of Hg; Barometer reading = 750 mm of Hg; Mean temperature of condensation = 35°C; Hot well temperature = 28°C; Mass of cooling water = 50,000 kg/h; Inlet temperature = 17°C; Outlet temperature = 30°C; Mass of condensate per hour = 1250 kg.
Find: (a) the mass of air present per m3 of condenser volume, (b) the state of steam entering the condenser and (c) the vacuum efficiency.
Take R for air = 287 J/kg K.
Solution
Given that vacuum = 690 mm of Hg; Barometer reading = 750 mm of Hg; tc = 35°C; th = 28°C; ṁw = 50000 kg/h; tf = 17°C; to = 30°C; ṁs = 1250 kg/h; R = 287 J/kg K
- We know that pressure in the condenser,pc = 750 − 690 = 60 mm of Hg = 60 × 0.00133= 0.08 barFrom steam tables, corresponding to a condensation temperature of 35°C, we find that the pressure of steam,ps = 0.05628 bar∴ Pressure of air (as per Dalton’s law),pa = pc − ps = 0.08 − 0.05628 = 0.02372 bar = 0.02372 × 105 = 2372 N/m2Mass of air per m3 of condenser volume,
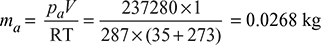
- Let x = Dryness fraction (i.e., state) of steam entering the condenser.From steam tables, corresponding to a pressure of 0.05628 bar (or 35°C), we find that,hf = 146.66 kJ/kg and hfg = 2418.6 kJ/kgCorresponding to a hot well temperature of 28°C,hf1 = 117.3 kJ/kgTotal heat of entering steam,h = hf + xhfg = 146.66 + x × 2418.6 kJ/kgMass of cooling water (mw).
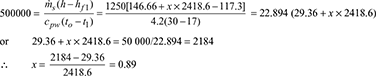
- Corresponding to a condensation temperature of 35°C,Ideal pressure of steam, = 0.05628 bar =
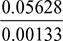 = 42.32 mm of Hg∴ Ideal vacuum = Barometer reading − Ideal pressure= 750 − 42.32 = 707.68 mm of HgVacuum efficiency, ηvac =
= 42.32 mm of Hg∴ Ideal vacuum = Barometer reading − Ideal pressure= 750 − 42.32 = 707.68 mm of HgVacuum efficiency, ηvac =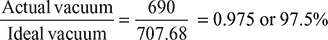
Example 8.23
The following observations were recorded during a test on a steam condenser:
Barometer reading = 765 mm of Hg
Condenser vacuum = 710 mm of Hg
Mean condenser temperature = 35°C
Condensate temperature = 28°C
Condensate collected per hour = 2 tonnes
Quantity of cooling water per hour = 60 tonnes
Temperature of cooling water at inlet = 10°C
Temperature of cooling water at outlet = 25°C
Find: (a) vacuum corrected to the standard barometer reading, (b) vacuum efficiency of the condenser, (c) undercooling of the condensate, (d) condenser efficiency, (e) quality of the steam entering of condenser, (f) mass of air per m3 of condenser volume and (g) mass of air per kg of uncondensed steam.
Solution
Given that Barometer reading = 765 mm of Hg; Condenser vacuum = 710 mm of Hg; T = 35°C = 35 + 273 = 308 K; tc = 28°C; ṁs = 2 t/h = 2000 kg/h; ṁw = 60 t/h = 60000 kg/h; ti = 10°C; to = 25°C
- Absolute pressure in the condenser = Barometer reading − Condenser vacuum= 765 − 710 = 55 mm of HgVacuum corrected to the standard barometer reading (assuming 760 mm of Hg)= 760 − 55 = 705 mm of Hg
- From steam tables, corresponding to the mean condenser temperature of 35°C, we find that ideal pressure of steam,
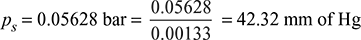 Ideal vacuum = Barometer pressure − Ideal pressure= 765 − 42.32 = 722.68 mm of HgVacuum efficiency, ηvac =
Ideal vacuum = Barometer pressure − Ideal pressure= 765 − 42.32 = 722.68 mm of HgVacuum efficiency, ηvac =  = 0.9824 or 98.24%
= 0.9824 or 98.24% - Undercooling of the condensate = Mean condenser temperature − Condensate temperature= 35 − 28 = 7°C
- Pressure in the condenser, pc = 765 − 710 = 55 mm of Hg= 55 × 0.00133 = 0.073 barFrom steam tables, corresponding to a pressure of 0.073 bar, we find that vacuum temperature,

- Let x = Quality of steam entering the condenser.From steam tables, corresponding to a pressure of 0.073 bar, we find that,hf = 166.7 kJ/kg; and hfg = 2407.4 kJ/kgCorresponding to a condensate temperature of 28°C, heat in condensate,hf1 = 117.3 kJ/kgTotal heat of entering steam,h = hf + xhfg = 166.7 + x × 2407.4Mass of cooling water (mw),
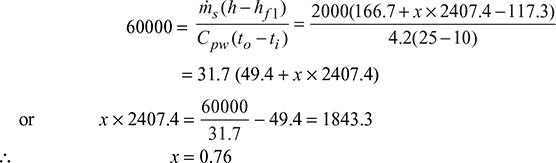
- Absolute pressure of air (as per Dalton’s law),pa = pc − ps = 0.073 − 0.0562 = 0.0168 bar= 0.0168 × 105 = 1680 N/m2∴ Mass of air per m3 of condenser volume,

- From steam tables, corresponding to 35°C (i.e., mean condenser temperature), specific volume of steam,vg = 25.245 m3/kgAir associated with 1 kg of steam at 35°C will occupy the same volume, i.e., 25.245 m3∴ Mass of air per kg of uncondensed steam,
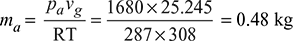
Example 8.24
The following observations refer to a surface condenser:
Mass flow rate of condensate = 20 kg/min
Mass flow rate of cooling water = 800 kg/min
Mean temperature of condensation = 35°C
Condenser vacuum = 0.954 bar
Barometer reading = 1.03248 bar
Intel cooling water temperature = 20°C
Outlet cooling water temperature = 30°C
Temperature of the hot well = 29°C
Calculate the following:
- Weight of air per unit volume of condenser
- Entering condition of steam to the condenser
- Vacuum efficiency of the condenser.
Properties of saturated steam:

Take R for air = 0.287 kJ/kg. K.
[IAS, 2009]
Solution
Given that ṁs = 20 kg/min, ṁw = 800 kg/min, T = 273 + 35 = 308 K, twi = 20°C, two = 30°C, ph = 1.03248 N/m2, pcu = 0.954 N/m2, thw = 29°C
Absolute pressure in condenser, pt = (ph − pcu) = 1.03248 − 0.954 = 0.07848 bar
At t = 35°C, ps = 0.05628
Partial pressure of air, pa = pt − ps = 0.07848 − 0.05628 = 0.0222 bar
- Mass of air, ma =
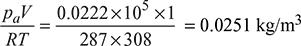
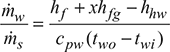 At pt = 0.07848 bar, hf = 169.15 kJ/kg, hfg = 2405.80 kJ.kg
At pt = 0.07848 bar, hf = 169.15 kJ/kg, hfg = 2405.80 kJ.kg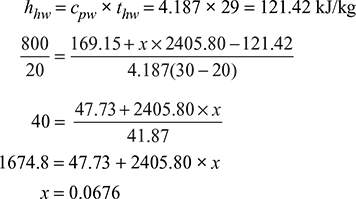
- Vacuum efficiency, ηvac =
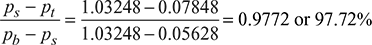
Leave a Reply