For any hydrocarbon fuel, we have
xCnH2n + 2 + yO2 → aCO2 + bH2O
where x, y, a, and b are constants to be evaluated.
Equating the weights of C, H, and O on both sides of the equation, we have
xn = a
or a = xn
x(2n + 2) = 2b
or b = [x/2] (2n + 2) = x(n + 1)
2y = 2a + b
or y = a + [b/2] = xn + [x/2] (n + 1) = [x/2] (3n + 1)
Substituting there values in combustion equation, we get
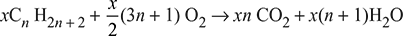
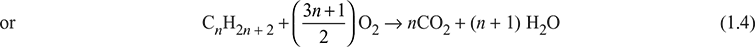
Leave a Reply