The ideal vapour absorption refrigeration system is shown in Fig. 19.22
Let Qg = heat given to the refrigerant in the generator.
Qrc = heat rejected from the condenses to the atmosphere.
Qra = heat rejected from the absorber to the atmosphere.
Qe = heat absorbed by the refrigerant in the evaporator.
Qp = heat added to the refrigerant due to pump work.
Qr = Qrc + Qra
= total heat rejected from the condenser and absorber to atmosphere
For heat balance, neglecting Qp according to the first law of thermodynamics, we have
Qr = Qrc + Qra = Qg + Qc
Let Tg = temperature at which heat is given to the generator.
Tr = temperature at which heat is rejected to atmosphere or cooling water from the condenser and absorber.
Te = temperature at which heat is absorbed in the evaporator.
The vapour absorption system can be considered as a perfectly reversible system. Therefore, the initial entropy of the system must be equal to the entropy of the system after the change in its conditions.
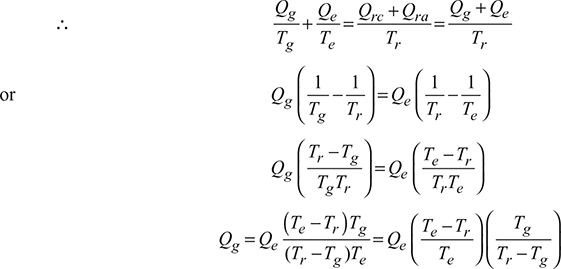
Maximum coefficient of performance,
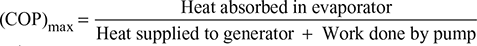
Neglecting pump work,
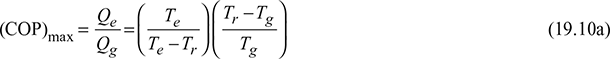

If Ta = temperature at which heat is rejected from the absorber
Tc = temperature at which heat is rejected from the condenser.
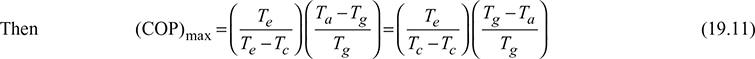
Example 19.13
A geothermal well at 130°C supplies heat at a rate of 100,500 kJ/h to an absorption refrigeration system. The environment is at 30°C and the refrigerated space is maintained at −22°C. Determine the maximum possible heat removal from the refrigerated space.
Solution
Given: Tr = 273 + 35 = 308 K, Tg = 273 + 110 = 383 K, Te = 273 − 5 = 268 K
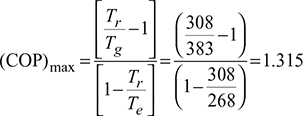
Example 19.14
In an absorption type refrigerator, the heat is supplied to ammonia generator by condensing steam at 2.5 bar and dryness fraction 0.9. The temperature in the refrigerator is to be maintained at −5°C. Calculate the maximum COP possible.
If the refrigeration load is 20 tonnes and actual COP is 75% of the maximum COP, find the mass of steam required per hour. Take ambient temperature as 30°C.
Solution
Given: p = 2.5 bar, x = 0.9, Te = 273 − 5 = 268 K, Q = 20TR, (COP)max = 0.75(COP)max, Tr = 273 + 30 = 303 K, From steam tables hf = 535.34 kJ/kg. hg = 2716.9 kJ/kg.
Saturation temperature of steam at p = 2.5 bar, Ts = 273 + 127.43 = 400.43 K = Tg
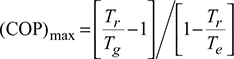
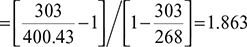
(COP)act = 0.75 × 1.863 = 1.397
Actual heat supplied = ![]()
Latent heat of steam, hfg = hg − hf = 2716.9 − 535.35 = 2181.56 kJ/kg
Mass of steam required per hour, ![]()
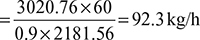
Leave a Reply