The air standard Brayton or Joule cycle is a constant pressure cycle used in gas turbine power plants. The p-v and T-S diagrams are shown in Fig 9.12. It consists of the following processes:
1−2: Isentropic compression in the compressor
2−3: Constant pressure heat addition.
3−4: Isentropic expansion of air
4−4: Constant pressure heat rejection
Consider 1 kg of working fluid
From first law of thermodynamics for steady flow, rejecting ΔKE and ΔPE, we have
δq – δw = dh
Heat added, qs = h3 – h2 = cp (T3 – T2)
Heat rejected qr = h4 – h1 = cp (T4 – T1)
Net work done by turbine, wnet = qs – qr = cp [(T3 − T2) − (T4 − T1)]
Also work done by turbine, wt = h3 − h4 = cp (T3 − T4)
Work consumed by compressor, wc= h2 − h1 = cp (T2 − T1)
wnet = cp [(T3 − T4) − (T2 − T1)] = cp [(T3 − T2) − (T4 − T1)]
Thermal efficiency,![]()
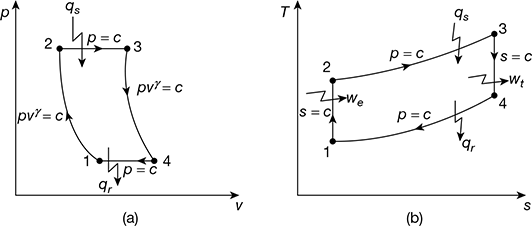
Figure 9.12 Brayton (or Joule) cycle: (a) p-v diagram, (b) T-s diagram
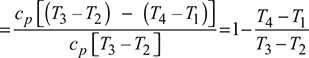
From isentropic compression process 1 – 2, we have
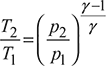
and from isentropic expansion process 3 – 4,
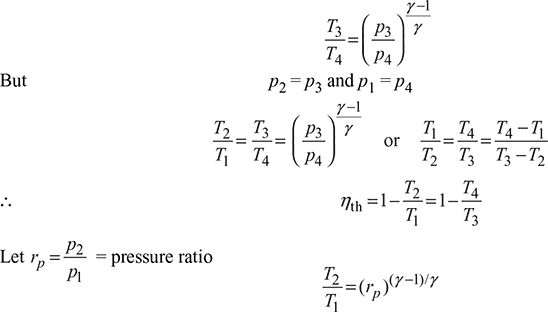
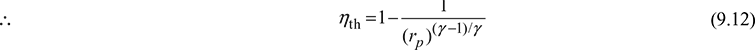
The variation of thermal efficiency v’s pressure ratio is shown in Fig. 9.13. The thermal efficiency increases with increasing values of pressure ratio. This cycle is used in gas turbines.

wt = work done by turbine
wc = work supplied to compressor
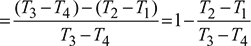
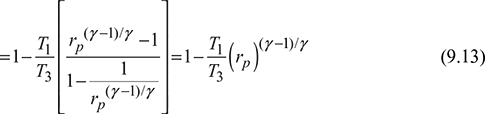
Pressure ratio for maximum work,
W = mcp [(T3 – T2) – (T4 – T1)]
= mcp [(T3 – T4) – (T2 – T1)]
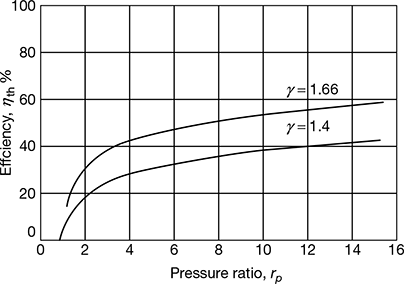
Figure 9.13 Efficiency v’s pressure ratio in simple Brayton cycle
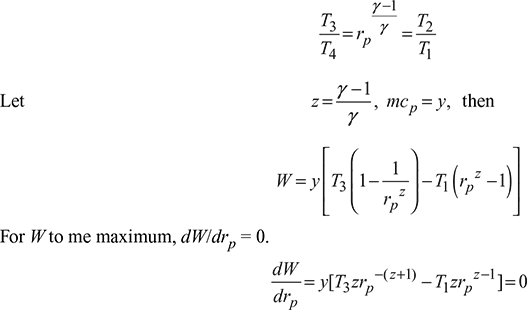
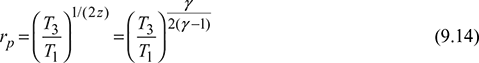
Leave a Reply