The refrigerants and their applications are given in Table 18.1.
The primary-refrigerants are further classified into the following four groups
- Halo-carbon refrigerants
- A zoetrope refrigerants.
- Inorganic refrigerants, and
- Hydro-carbon refrigerants.
- Halo-carbon Refrigerants: The commonly used halo-carbon refrigerants, their applications and method of leak testing are given in Table 18.2.
- Azeotrope Refrigerants: The term ‘azeotrope’ refers to a stable mixture of refrigerants whose vapour and liquid phases retain identical compositions over a wide range of temperatures.Some of the azeotropic refrigerants, their applications and method of leak testing are given in Table 18.3.
Table 18.1 Refrigerants and their applications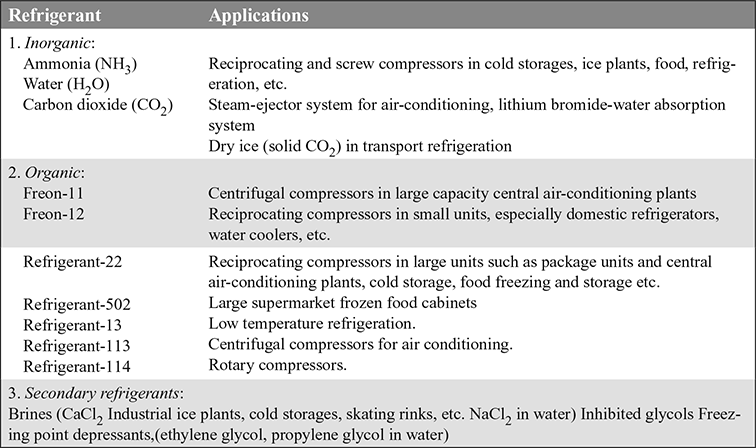
Table 18.2 Commonly used halo carbon refrigerants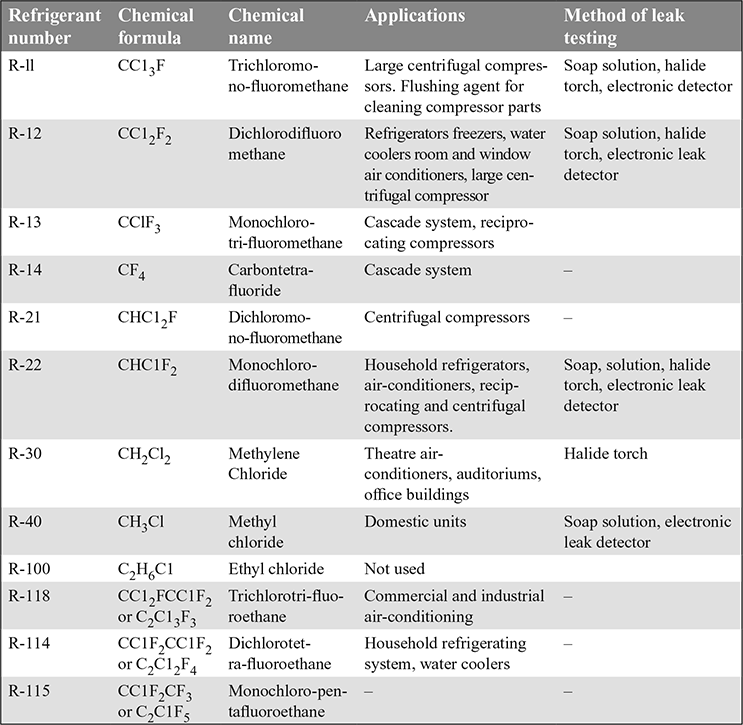
Table 18.3 Azeotrope refrigerantss

- 3. Inorganic Refrigerants: The various inorganic refrigerants, their applications and leak testing method are given in Table 18.4.
- 4. Hydro-Carbon Refrigerants: The various hydro-carbons are given in Table 18.5. They are mostly used for industrial and commercial installations.
Table 18.4 Inorganic refrigerants
Table 18.5 Hydrocarbon refrigerants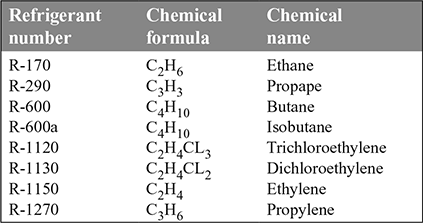
Table 18.6 Ozone Depletion Potential (ODP) of Refrigerants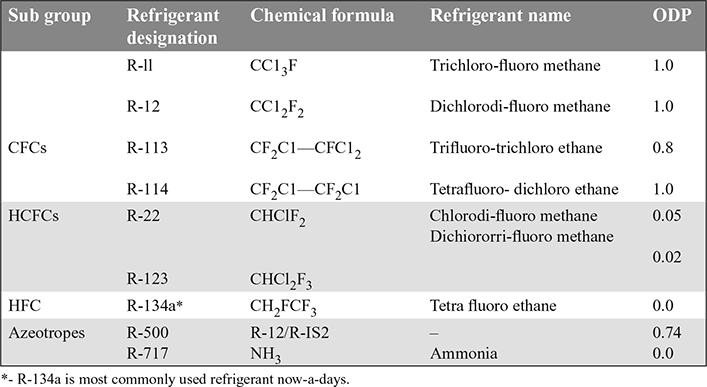
- 5. Ozone Layer Depletion: Stratosphere is about 25-30 km away from the earth’s surface. It contains ozone (O3). Ozone is formed in the stratosphere by the reaction that occurs when sunlight interacts with oxygen. This ozone shelters the earth from ultraviolet (UV) radiation. This protection is essential for human health. This ozone is easily destroyed by the UV radiation. The sun’s radiation will break down the O3 molecule into a standard O2 molecule and elemental free oxygen (O) atom. More O3 is produced through photosynthesis and the bonding of O2 with free oxygen O. Thus ozone is constantly being formed and destroyed in the stratosphere.In the stratosphere, UV radiation will break off a chlorine atom (Cl) from the CFC and HCFC molecule. The chlorine atom destroys many O3 molecules The depleted O3 molecules in the stratosphere let more UV radiation reach the earth This causes an increase in skin cancers and frequency of cataracts in humans; weakens human immune system and decreases plant and marine life. Ozone depletion potential (ODP) has been used as an index to show the effect of a refrigerant on O3. Lower the ODP, it will be better. The ODP of some of the refrigerants is given in Table 18.6.
Leave a Reply