For an adiabatic compression process, there is no heat exchange with the surroundings. If the adiabatic process is reversible (frictionless), then the process is called isentropic process in which the entropy of the system does not change. However, in an actual compressor, during adiabatic compression, there are losses due to friction in air and blade passages, eddies formation, and shocks at entry and exit. These factors cause internal heat generation and consequently, the maximum temperature reached would be higher than that for adiabatic compression. This results in a progressive increase in entropy. Such a process, although adiabatic, is not reversible adiabatic or isentropic.
The isentropic and adiabatic processes for the static and stagnation values are shown in Fig. 14.5 on the T-s diagram. Process 1-2 is the isentropic process for the static temperature and 01-02 for the total head temperature. Processes 1-2′ and 01-02′ are the adiabatic processes for the static and total head temperatures, respectively.
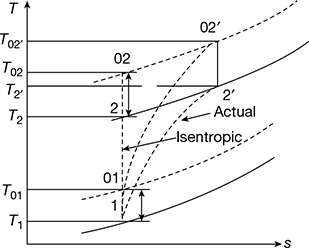
Figure 14.5 Isentropic and actual compression
Leave a Reply