Thermodynamics has its own unique vocabulary associated with it. A good understanding of the basic concepts forms a sound understanding of various topics discussed in thermodynamics preventing possible misunderstandings.
Thermodynamic Systems
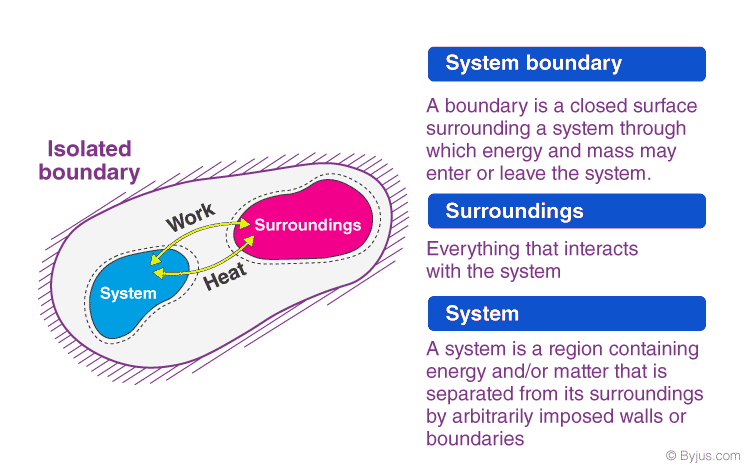
System
A thermodynamic system is a specific portion of matter with a definite boundary on which our attention is focused. The system boundary may be real or imaginary, fixed or deformable.
There are three types of systems:
- Isolated System – An isolated system cannot exchange energy and mass with its surroundings. The universe is considered an isolated system.
- Closed System – Across the boundary of the closed system, the transfer of energy takes place but the transfer of mass doesn’t take place. Refrigerator, compression of gas in the piston-cylinder assembly are examples of closed systems.
- Open System – In an open system, the mass and energy both may be transferred between the system and surroundings. A steam turbine is an example of an open system.
| Interactions of thermodynamic systems | |||
| Type of system | Mass flow | Work | Heat |
| Isolated System | ☓ | ☓ | ☓ |
| Open System | ✓ | ✓ | ✓ |
| Closed System | ☓ | ✓ | ✓ |
Surrounding
Everything outside the system that has a direct influence on the behaviour of the system is known as a surrounding.
Leave a Reply