Eq. (20.16) can be written as,
h= (cpa + wcpv)td + w(hfg)0°C

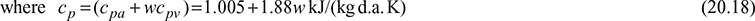
is termed as the humid specific heat. It is the specific heal of moist air (1 + w) kg per kg of dry air.
At low temperatures of air-conditioning the value of w is very small. An approximate value of cp of 1.0216 kJ/(kg d.a.).K may be taken for nil practical purposes
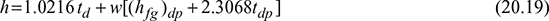
Example 20.1
A mixture of dry air and water vapour is at a temperature of 20°C under a total pressure of 735 mm Hg. The dew point temperature is 15°C. Calculate the following:
- Partial pressure of water vapour,
- Relative humidity,
- Specific humidity,
- Specific enthalpy of water vapour
- Enthalpy of air per kg of dry air, and
- Specific volume of air per kg of dry air.
Solution
- From steam tables, the partial pressure of water vapour at tdp = 15°Cpv =12.79mm HgNow 760mm Hg = 1.01325×105 N/m21mm Hg = 133.32 N/m2∴ pv =12.79×133.32=1705.16N/m2
- Saturation pressure of water vapour at 20°C DBTps =17.54mm Hg = 17.54 × 133.32 = 2338.4 N/m2Relative humidity,
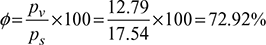
- Specific humidity, w =
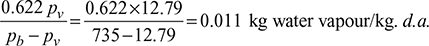
- Latent heat of vaporization of water at td = 20°C, (hfg)20°C = 2454.1kJ/kgLatent heat of vaporization of water at tdp =15°C, (hfg)15°C = 2465.9 kJ/kgSpecific enthalpy of water vapour,hv =[4.1868tdp + (hfg)dp +1.88(td − tdp)]=[4.1868×15+ 2465.9+1.88(20 −15)]= 2538.1 kJ/kg w.v
- Enthalpy of air per kg d.a., h = 1.005 td + whv= 1.005 × 20 + 0.011 × 2538.1= 48.02 kJ/kg d.a.
- Specific volume of air per kg of d.a.
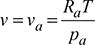
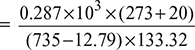 = 0.873 m3/kg d.a
= 0.873 m3/kg d.a
Example 20.2
A sling psychrometer gave the following readings:
DBT = 30°C, WBT = 20°C, Barometer reading = 740 mm of Hg.
Determine: (a) dew point temperature, (b) relative humidity, (c) specific humidity (d) degree of saturation, (e) vapour density, and (f) enthalpy of mixture per kg of dry air
Solution
Given: td = 30°C, tw = 20°C, pb = 740 mm Hg
- Corresponding to tw = 20°C, from steam tables, saturation pressure, pw = 0.023385 barBarometric pressure, pb = 740 × 133.32 × 10−5 = 0.986568 barPartial pressure of water vapour
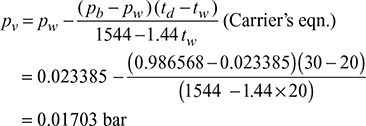 Corresponding to pv = 0.01703 bar pressure, from steam tables, dew point temperaturetdp = 15°C
Corresponding to pv = 0.01703 bar pressure, from steam tables, dew point temperaturetdp = 15°C - Corresponding to td = 30°C, from steam tables, saturation pressure of water vapour,ps = 0.04246 bar
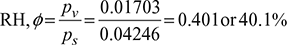
- Specific humidity,
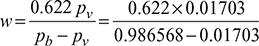 = 0.010925 kg/kg d.a
= 0.010925 kg/kg d.a - Specific humidity of saturated air,
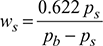
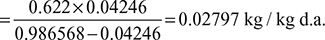 Degree of saturation,
Degree of saturation,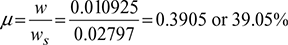
- Vapour density,
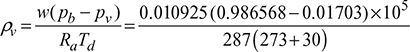 = 0.01218 kg/m3 of d.a.
= 0.01218 kg/m3 of d.a. - From steam tables, (hfg)dp = 15°C = 2465.9 kJ/kgEnthalpy of mixture per kg d.a.,h = 1.0216 td + w[(hfg)dp + 2.3068 tdp]= 1.0216 × 30 + 0.010925 [2465.9 + 2.3068 × 15] = 57.966 kJ/kg d.a.
Example 20.3
A room 5m × 4m × 3m contains moist air at 40°C. The barometric pressure is 1 bar and relative humidity is 70%. Determine: (a) humidity ratio, (b) dew point temperature, (c) mass of dry air, and (d) mass of water vapour.
If the moist air is further cooled at constant pressure until the temperature is 15°C, find the amount of water vapour condensed.
Solution
Given: V = 5 × 4 × 3 = 60 m3, td = 40°C, pb = 1 bar, ϕ = 0.70.
- Saturation pressure of vapour corresponding to td = 40°C, from steam tables is,ps = 7.384 kPa or 0.07384 bar
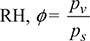 pv = 0.7× 0.07384 = 0.05169 barHumidity ratio,
pv = 0.7× 0.07384 = 0.05169 barHumidity ratio,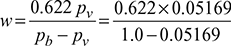 = 0.0339 kg/kg d.a or 33.9 g/kg d.a
= 0.0339 kg/kg d.a or 33.9 g/kg d.a - DPT, tdp = saturation temperature corresponding to pv = 0.05169 bar or 5.169 kPa= 33.46°C from steam tables
- pa = pb − pv = 1.0 − 0.05169 = 0.94831 barMass of dry air,
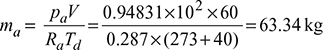
- Now w =
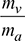 Mass of water vapour, mv = 0.0339 × 63.34 = 2.147 kgSaturation pressure corresponding to 15°C, from steam tables is,ps = pv = 1.707 kPa or 0.01707 bar
Mass of water vapour, mv = 0.0339 × 63.34 = 2.147 kgSaturation pressure corresponding to 15°C, from steam tables is,ps = pv = 1.707 kPa or 0.01707 bar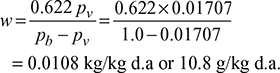 Now pa = pb − pv = 1 − 0.01707 = 0.98293 bar or 98.293 kPaMass of dry air,
Now pa = pb − pv = 1 − 0.01707 = 0.98293 bar or 98.293 kPaMass of dry air, 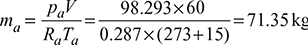 Mass of water vapour, mv = wma = 0.0108 × 71.35 = 0.77 kgAmount of water vapour condensed = 2.147 − 0.77 = 1.377 kg
Mass of water vapour, mv = wma = 0.0108 × 71.35 = 0.77 kgAmount of water vapour condensed = 2.147 − 0.77 = 1.377 kg
Leave a Reply