A catalyst is a substance that accelerates a chemical reaction by lowering the energy needed for it to proceed. It is not consumed in the reaction. A three-way catalytic converter reduces the concentration of CO, HC, and NOx in the exhaust. A catalytic converter is usually a stainless steel container mounted along the exhaust pipe of the engine. There is a porous ceramic (Al2O3) structure inside the container through which the exhaust gas flows. The ceramic structure is a single honey-comb structure with many flow passages. The ceramic passages contain small embedded particles of catalytic material such as platinum, palladium, rhodium, and so on that promote oxidation or reduction reaction in the exhaust gas. Platinum and palladium promote the oxidation of CO and HC, whereas rhodium promotes the reaction of NOx by reduction process.
The principle of working of a three-way catalytic converter is shown in Fig. 11.29(a) and (b).
1 Function of a Catalyst in a Catalytic Converter
The catalyst controls the level of various exhaust pollutants from the engine by changing the chemical characteristics of the exhaust gases. Catalyst materials such as platinum or platinum, palladium, and rhodium are used in the converter. CO and HC oxidise the CO2 and H2O by palladium and platinum and NOx is reduced by rhodium. An oxidation catalyst is placed downstream of the reduction catalyst.
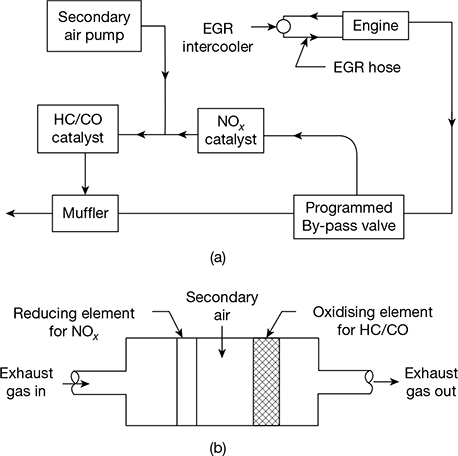
Figure 11.29 Three-way catalytic converter: (a) Catalytic converter package, (b) Three way catalytic converter
where z = x + 0.25 y
| Reduction | Reactions |
|---|---|
| 2NO + 2CO | ⇒ N2 + 2CO2 |
| 2NO + 5CO + 3H2O | ⇒ 2NH3 + 5CO2 |
| 2NO + CO | ⇒ N2O + CO2 |
| 2NO + 2H2 | ⇒ N2 + 2H2O |
| 2NO + 5H2 | ⇒ 2NH3 + 2H2O |
| 2NO + H2 | ⇒ N2O + H2O |
Leave a Reply