This cycle has two reversible adiabatics (isentropics), one isobar and one isochore. The p-v and T-s diagrams are shown in Fig. 9.5. For one kg of the working fluid, we have
Heat supplied, qs = cv (T3 – T2)
Heat rejected, qr = cp (T4 – T1)
Net work done, wnet = qs − qr = cv (T3 −T2) − cp (T4 −T1)
Thermal efficiency, 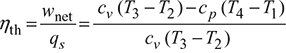
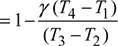
where ![]()
Let compression ratio, ![]()
Explosion ratio, ![]()
From compression process 1 − 2, we have
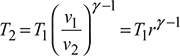
For constant volume heat addition process 2 − 3, we have
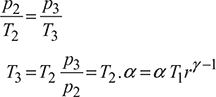
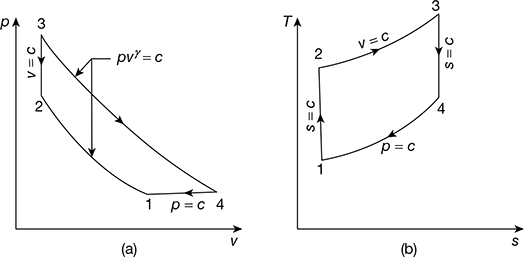
Figure 9.5 Atkinson cycle: (a) p-v diagram, (b) T-s diagram
From expansion process 3−4, we have
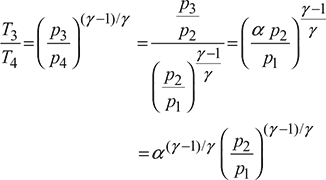


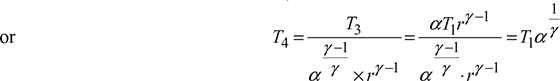
Substituting the values of T2, T3 and T4, we get
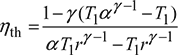
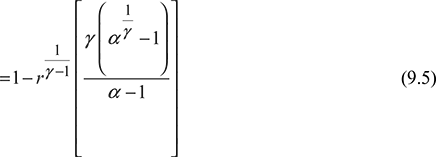
Leave a Reply