Let ṁw = mass flow rate of circulating water, kg/h
ṁs = mass flow rate of steam condensed, kg/h
t = temperature of wet exhaust steam, °C
tc = temperature of condensate, °C
hf = enthalpy of water at t°C, kJ/kg
hfg = enthalpy of evaporation of steam at t°C, kJ/kg
x = dryness fraction of exhaust steam
tiw = inlet temperature of cooling water, °C
tow = outlet temperature of cooling water, °C
hc = enthalpy of condensate, kJ/kg = cpw tc
cpw = specific heat of cooling water, 4.1868 kJ/kg.K
Now Heat given by steam = Heat absorbed by water
For jet condenser,
tow = tc

For surface condenser,
ms (hf + xhfg − hc) = mw cpw (tow − tiw)

Example 8.2
A 200 kW steam engine has a steam consumption of 10 kg/kWh. The back pressure of the engine is equal to the condenser pressure of 0.15 bar. The condensate temperature is 32°C. The cooling water temperature at inlet and outlet are 20°C and 30°C, respectively. Calculate the mass of cooling water required per hour if the exhaust steam is dry saturated.
Solution
At 0.15 bar, hf = 225.91 kJ/kg, hfg = 2373.1 kJ/kg, tc = 32°C, tow = 30°C, tiw = 20°C
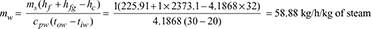
Total steam used per hour = 200 × 10 = 2000 kg
Total circulating water used = 58.88 × 2000 = 117760 kg/h
Leave a Reply