Determine the condition of steam in the following cases:
- At a pressure of 10 bar and temperature 200°C.
- At a pressure of 10 bar and specific volume 0.175 m3/kg.
Solution
Given p = 10 bar; t = 200°C; v = 0.175 m3/kg
- Condition of steam at temperature of 200°C:From steam table (A.1.2 in the Appendix), corresponding to a pressure of 10 bar, we find the saturation temperature ts = 179.91°CSince the saturation temperature at 10 bar is (179.91°C) lower than the given temperature of the steam (200°C), therefore the given steam is superheated.The degree of superheat = 200 − 179.91= 20.09°C
- Condition of steam at a volume of 0.175 m3/kg:From steam table (A.1.2 in the Appendix), vf = 0.001127 m3/kg, vg = 0.19444 m3/kg.Since the volume of given steam (0.175 m3/kg) is less than the specific volume of the dry saturated steam (0.19444 m3/kg), therefore the given steam is wet. Therefore, the dryness fraction can be found to be 0.175 = 0.001127 + x (0.19444 − 0.001127)or x = 0.8994
Example 2.2
Calculate the quantity of heat required to generate 1 kg of steam at a pressure of 8 bar from water at 30°C (a) when dryness fraction is 0.9, (b) when steam is just dry, and (c) when the temperature of steam is 300°C.
From steam tables (A.1.2 in the appendix) at 8 bar: hf = 721.1 kJ/kg, hfg = 2048.0 kJ/kg
And at 30°C (A.1.1 in the appendix), hf = 125.79 kJ/kg
- h = hf + x hfg = 721.1 + 0.9 × 2048.0 = 2564.3 kJ/kgNet heat required = h − hf = 2564.3 − 125.79 = 2438.51 kJ/kg
- h = hf + hfg = hg = 2769.1 kJ/kgNet heat required = h − hf = 2769.1 − 125.79 = 2643.31 kJ/kg
- ts = 170.4°C, tsup = 300°C
Therefore, the steam is in superheated condition. From superheated steam table (A.1.3 in the appendix), we have h = 3056.4 kJ/kg
Net heat required = h − hf = 3056.4 − 125.77 = 2930.63 kJ/kg
Example 2.3
A pressure cooker contains 2 kg of steam at 5 bar and 0.9 dry. Find the quantity of heat that must be rejected so that the quality of steam becomes 0.5 dry.
Solution
The pressure cooker is a constant volume vessel. Therefore, W = 0
∴ Q = ∆U = U2 − U1
From steam tables (A.1.2 in the appendix) at 5 bar: vf = 0.001093 m3/kg, vg= 0.3749 m3/kg, uf = 639.66 kJ/kg, ufg = 1921.6 kJ/kg,
u1 = uf + x1ufg = 639.66 + 0.9 × 1921.6 = 2369.1 kJ/kg
U1 = 2 × 2369.1 = 4738.2 kJ
Specific volume of the steam at 5 bar
v1 = vf + x1 (vg − vf) = 0.001093 + 0.9 × (0.3749 − 0.001093) = 0.3375 m3/kg
From steam tables (A.1.2 in the appendix), the properties of steam at 2.5 and 2.75 bar are given below:
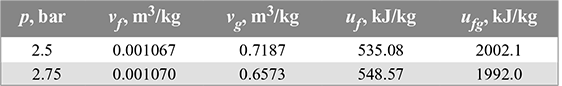
For constant volume of steam, we have
v1 = v2 = 0.3375 m3/kg
Specific volume of the steam at 2.5 bar
v = vf + x (vg − vf ) = 0.001067 + 0.9 × (0.7187 − 0.001067) = 0.3599 m3/kg
Specific volume of the steam at 2.75 bar
v = vf + x(vg − vf ) = 0.001070 + 0.9 × (0.6573 − 0.001070) = 0.3292 m3/kg
Using linear interpolation, the pressure corresponding to v2 = 0.3375 m3/kg is obtained as
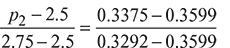
or p2 = 2.68 bar
Using linear interpolation, we get
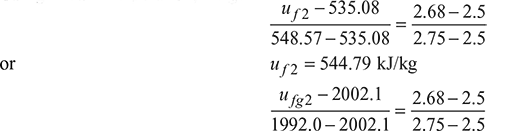
or u fg2 = 1994.83 kJ/kg
∴ u2 = uf 2 + x2 ufg2 = 544.79 + 0.5 × 1994.83 = 1542.205 kJ/kg
U2 = m u2 = 2 × 1542.205 = 3084.41 kJ
Q = U2 − U1 = 3084.41 − 4738.2 = − 1653.79 kJ
Example 2.4
About 5400 m3/h of wet steam with dryness fraction 0.92 and at 10 bar is to be supplied by a boiler for a processing plant. Calculate (a) the mass of steam supplied per hour and (b) the quantity of coal of calorific value 15 MJ/kg to be burnt in the boiler if overall efficiency of the boiler is 30%.
Solution
- From steam tables (A.1.2 in the appendix) at 10 bar: vf = 0.001127 m3/kg, vg= 0.19444 m3/kgSpecific volume of the steam v = vf + x (vg − vf ) = 0.001127 + 0.92 × (0.19444 − 0.001127) = 0.179 m3/kg
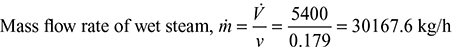
- At p = 10 bar, hf = 762.79 kJ/kg, hfg = 2015.3 kJ/kgh = hf + x hfg = 762.79 + 0.92 × 2015.3 = 2616.866 kJ/kgH = ṁh = 30167.6 × 2616.866 = 78944566 kJ/hHeat supplied to boiler, Hb =
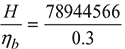 = 263148553 kJ/hQuantity of coal burnt =
= 263148553 kJ/hQuantity of coal burnt = 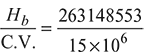 = 17.54 tonnes/h
= 17.54 tonnes/h
Example 2.5
Find the internal energy of 1 kg of superheated steam at a pressure of 10 bar and 300°C. If this steam is expanded to 1.5 bar and 0.9 dryness, then find the change in internal energy.
Solution
From steam table (Appendix A.1.3), at p = 10 bar and tsup = 300°C, for superheated steam,
u1 = 2793.2 kJ/kg
For dry saturated steam, at p = 1.5 bar, we have from table A.1.2 in the Appendix
uf = 466.92 kJ/kg, ufg = 2052.7 kJ/kg,
u2 = uf 2 + x2ufg2 = 466.92 + 0.9 × 2052.7 = 2314.35 kJ/kg
∆u = u2 − u1 = 2314.35 − 2793.2 = − 478.85 kJ/kg
Example 2.6
A rigid vessel is initially divided into two parts A and B by a thin partition. Part A contains 1 kg of steam at 4 bar, dry and saturated, and part B contains 2 kg of steam at 8 bar with dryness fraction of 0.90. The partition is removed and the pressure in the vessel after sometimes is found to be 6 bar. Find (a) the volume of the vessel and the state of steam and (b) the amount of heat transfer from steam to the vessel and the surroundings.
Solution
- At pA = 4 bar, from steam table A.1.2 in the Appendix, vgA = 0.4625 m3/kg, uA = 2553.6 kJ/kgVA = mAvgA = 1 × 0.4625 = 0.4625 m3At pB = 8 bar, from steam table A.1.2 in the Appendix, vfB = 0.001115 m3/kg, vgB = 0.2404 m3/kgVB = mB (vf B + xBvgB) = 2 × [0.001115 + 0.9 × (0.2404 − 0.001115)] = 0.433 m3Volume of vessel, V = VA + VB = 0.4625 + 0.433 = 0.8955 m3At p = 6 bar, from steam table A.1.2 in the Appendix, vf = 0.001101 m3/kg, vg = 0.3157 m3/kg,m = 1 + 2 = 3 kgDryness fraction at 6 bar, x can be found to be
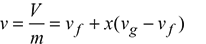
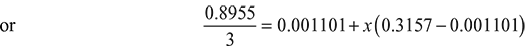 or x = 0.945
or x = 0.945 - Now UA = mAuA = 1 × 2553.6 = 2553.6 kJUB = mB (hfB + xBhfgB − pBxBvgB × 102)= 2 (721.1 + 0.9 × 2048.0 − 8 × 0.9 × 0.240 × 102)= 4783.0 kJAt 6 bar, U = m[hf + x hfg − pxvg × 102]= 3[670.6 + 0.944 × 2086.3 − 6 × 0.944 × 0.316 × 102)= 7383.25 kJQ = U − (UA + UB)= 7383.25 − (2553.3 + 4783.0) = 46.95 kJ
Example 2.7
Calculate enthalpy and entropy for 5 kg of steam at 8 bar under the following conditions:
(a) dry and saturated, (b) wet steam having wetness of 40%, and (c) superheated steam at 250°C.
Solution
From steam table (Appendix A.1.2), at p = 8 bar, we have
hf = 721.1 kJ/kg, hfg = 2048.0 kJ/kg, hg = 2769.1 kJ/kg, sf = 2.0461 kJ/kgK, sfg = 4.6166 kJ/kgK, sg = 6.6627 kJ/kgK
At tsup = 250°C and p = 8 bar, we have from table A.1.3
h = 2950.0 kJ/kg, s = 7.0384 kJ/kgK
- H = m hg = 5 × 2769.1 = 13845.5 kJS = m sg= 5× 6.6627 = 33.3135 kJ/K
- x = 1 − 0.4 = 0.6H = m (hf + x hfg) = 5(721.1 + 0.6 × 2048.0) = 9749.5 kJS = m (sf + xsfg) = 5(2.0461 + 0.6 × 4.6166) = 24.08 kJ/K
- H = mh = 5 × 2950.0 = 14750.0 kJS = ms = 5 × 7.0384 = 35.192 kJ/K
Example 2.8
About 1 kg of steam 0.85 dry at a pressure of 1 bar and dryness fraction 0.85 is compressed in a cylinder to a pressure of 2 bar according to pv1.25 = const. Determine the final condition of steam and the heat transfer through the cylinder walls.
Solution
From steam table (Appendix A.1.2), at p = 1 bar, vf1 = 0.001043 m3/kg, vg1 = 1.694 m3/kg
uf1 = 417.33 kJ/kg, ufg1 = 2088.7 kJ/kg
Specific volume of steam
v = vf + x (vg − vf) = 0.001043 + 0.85 × (1.694 − 0.001043) = 1.44 m3/kg
Now p1v1n = p2v2n
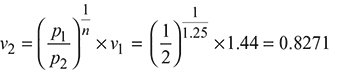
At p2 = 2 bar, vf 2 = 0.001061 m3/kg, vg2 = 0.8857 m3/kg, uf 2 = 504.47 kJ/kg, ufg2 = 2025 kJ/kg
Now v2 = 0.8271 = vf 2+ x2 (vg2 − vf 2) = 0.001061 + x2 (0.8857− 0.001061)
or x2 = 0.934
Now u1 = uf 1 + x1ufg1 = 417.33 + 0.85 × 2088.7 = 2192.72 kJ/kg
u2 = uf 2 + x2ufg2 = 504.47 + 0.934 × 2025 = 2395.82 kJ/kg
∆u = u2 − u1 = 2395.82 − 2192.72 = 203.1 kJ/kg
Work done,
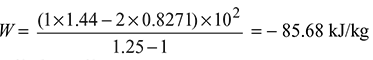
Heat transfer through the cylinder walls,
q = w + (u2 − u1) = − 85.68 + 203.1 = 117.42 kJ/kg
Example 2.9
Steam at a pressure of 10 bar and 0.9 dryness fraction expands to 1 bar hyperbolically. Find (a) work done and heat absorbed and (b) internal energy and change in enthalpy.
Take specific heat of steam at constant pressure = 2 kJ/kg.K.
From steam table (Appendix A.1.2), we have
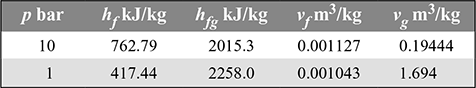
v1 = vf 1+ x1 (vg1 − vf 1) = 0.001127 + 0.9 × (0.19444 − 0.001127) = 0.1751 m3/kg
Now, p1v1 = p2v2
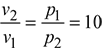
Expansion ratio, ![]()
Work done, w = p1v1 ln r = 10 × 102 × 0.1751 ln 10 = 403.18 kJ/kg
v2 = 10v1 = 10 × 0.1751 = 1.751 m3/kg
Now, v2 > vg2. Therefore, the steam is superheated at p2 = 1 bar.
Using linear interpolation, we have
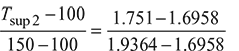
Tsup 2 = 111.47°C
h1 = hf 1 + x1hfg1 = 762.79 + 0.9 × 2015.3 = 2576.56 kJ/kg
Using linear interpolation, we get
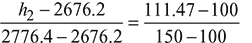
h2 = 2699.19 kJ/kg
∆h = h2 − h1 = 2699.19 − 2576.56 = 122.63 kJ/kg
∆u = ∆h for hyperbolic process
q = w + ∆u = 403.18 + 122.63 = 525.81 kJ/kg
Example 2.10
Steam, which is initially dry saturated, expands isentropically from pressure of 15 bar to 0.15 bar. Find the index of isentropic expansion.
Solution
From steam table (Appendix A.1.2), we have

For isentropic expansion, s1 = s2
s1 = sf 1 + sfg1 = 2.315 + 4.130 = 6.445 kJ/kg.K
s2 = sf 2 + x2sfg2 = 0.7548 + x2 × 7.2536 = 6.445
or x2 = 0.7845
v1 = vg1 = 0.13177 m3/kg
v2 = vf 2+ x2 (vg2 − vf 2) = 0.001014 + 0.7845 × (10.022 − 0.001014) = 7.8625 m3/kg
Now, p1v1n = p2v2n
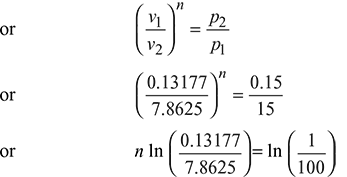
or n = 1.126
Example 2.11
Steam at a pressure of 8 bar and 0.95 dry is expanded to a pressure of 1.8 bar. Find the final condition of steam and the heat drop for each of the following methods of expansion:
- Adiabatic expansion
- Throttling expansion
Solution
Given that p1 = 8 bar, x1 = 0.95, p2 = 1.75 bar
From steam tables (Appendix A.1.2), at 8 bar, sf1 = 2.0461 kJ/kg.K, sfg1 = 4.6166 kJ/kg.K, hf1 = 721.1 kJ/kg, hfg1 = 2048 kJ/kg, and at 1.75 bar, sf2 = 1.4848 kJ/kg.K, sfg2 = 5.6868 kJ/kg.K, hf 2 =486.97 kJ/kg, hfg2 = 2213.6 kJ/kg
- During an adiabatic expansion, s1 = s2sf 1 + x1sfg1 = sf 2 + x2sfg22.0461 + 0.95 × 4.6166 = 1.4848 + x2 × 5.6868x2 = 0.8699
- During throttling process, h1 = h2hf 1 + x1hfg1 = hf 2 + x2hfg2721.1 + 0.95 × 2048.0 = 486.97 + x2 × 2213.6x2 = 0.9847
Example 2.12
Steam at 10 bar flows into a barrel containing 150 kg of water at 10°C. The final temperature of water is 25°C and the increase in mass is 4 kg. Calculate the dryness fraction of steam. Take cpw = 4.187 kJ/kg.K.
Solution
Given that ms = 4 kg, p = 10 bar, mw = 150 kg, tw1 = 10°C, tw2 = 25°C, and cps = 4.187 kJ/kg.K
From steam table (Appendix A.1.2), at 10 bar, hfg = 2015.3 kJ/kg and ts = 179.91°C
Heat lost by steam = Heat gained by water
ms[x hfg + cpw(ts − tw2)] = mw (tw2 − tw1)cpw
4[x × 2015.3 + 4.187(179.91 − 25)] = 150 (25 − 10) × 4.187
Dryness fraction of steam, x = 0.847
Example 2.13
In a separating and throttling calorimeter, the steam main pressure is 8 bar absolute and the temperature after throttling is 130°C. The pressure in the throttling calorimeter is 0.001 bar gauge, and the barometer reading is 75 cm. If 0.15 kg of water is trapped in the separator and 1.8 kg of steam is passed through the throttling calorimeter, then determine the dryness fraction of steam in the steam main.
Solution
After throttling, p2 = 0.001 + 0.75 × 9.81 × 13600 × 10−5 = 1.01 bar
The saturation temperature for p2 = 1.01 bar, ts2 = 99.62°C
Degree of superheat = tsup − ts2 = 130 − 99.62 = 30.38°C
For superheated steam, from steam tables (Appendix A.1.3), for tsup = 130°C
p = 1.01 bar, we have by linear interpolation
hsup2 = 2736.32 kJ/kg
From steam tables (Appendix A.1.2),
At 8 bar hf1 = 721.1 kJ/kg, hfg1 = 2048 kJ/kg
h1 = hf 1 + x1hfg1
Now h1 = hsup2
721.1 + x1 + 2048.0 = 2736.32
x1 = 0.984
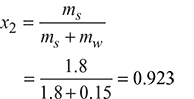
Dryness fraction, x = x1x2
= 0.984 × 0.923 = 0.908
Example 2.14
Calculate the enthalpy of 1 kg of steam at a pressure of 8 bar and dryness fraction of 0.8. How much heat would be required to raise 2 kg of this steam from water at 20°C?
Solution
Given that ms = 1 kg, p = 8 bar, x = 0.8, and tw = 20°C
Enthalpy of 1 kg of steam:
From steam table (Appendix A.1.2), corresponding to a pressure of 8 bar, we find that,
hf = 721.1 kJ/kg and hfg = 2048 kJ/kg
Enthalpy of 1 kg of wet steam,
h = hf + x hfg = 721.1 + 0.8 × 2048 = 2359.5 kJ
Heat required to raise 2 kg of this steam from water at 20°C:
We have calculated above the enthalpy or total heat required to raise 1 kg of steam from water at 0°C. Since the water, in this case, is already at 20°C, therefore,
Heat already in water = 4.2 × 20 = 84 kJ
Heat required per kg of steam = 2359.5 − 84 = 2275.5 kJ
and heat required for 2 kg of steam = 2 × 2275.5 = 4551 kJ
Example 2.15
Determine the quantity of heat required to produce 1 kg of steam at a pressure of 6 bar at a temperature of 25°C, under the following conditions:
- When the steam is wet having a dryness fraction 0.9
- When the steam is dry saturated
- When it is superheated at a constant pressure at 250°C, assuming the mean specific heat of superheated steam to be 2.3 kJ/kg.K.
Solution
Given that p = 6 bar, tw = 25°C, x = 0.9, tsup = 250°C, and cps = 2.3 kJ/kg.K
From steam table (Appendix A.1.2), corresponding to a pressure of 6 bar, we find that
hf = 670.4 kJ/kg, hfg = 2086.3 kJ/kg, and ts = 158.85°C
- When the steam is wet:Enthalpy or total heat of 1 kg of wet steam,h = hf + xhfg = 670.54 + 0.9 × 2086.3 = 2548.21 kJSince the water is at a temperature of 25°C, therefore from steam tables (Appendix A.1.1)Enthalpy of water 104.87 kJ∴ Heat actually required = 2548.21 − 104.87 = 2443.34 kJ
- When the steam is dry saturated:From steam tables (Appendix A.1.2), enthalpy or total heat of 1 kg of dry saturated steam,hg = 2756.8 kJ∴ Heat actually required = 2756.8 − 104.87 = 2651.93 kJ
- When the steam is superheated:From steam tables (Appendix A.1.3), enthalpy or total heat of 1 kg of superheated steam,hsup = 2957.2 kJ∴ Heat actually required = 2957.2 − 104.87 = 2852.33 kJ
Example 2.16
Steam enters an engine at a pressure of 12 bar with a 67°C of superheat. It is exhausted at a pressure of 0.15 bar and 0.95 dry. Find the drop in enthalpy of the steam. Assume specific heat of superheated steam cps = 2.1 kJ/kgK.
Solution
Given that p1 = 12 bar, tsup − ts = 67°C, p2 = 0.15 bar, and x = 0.95
From steam table (Appendix A.1.2), corresponding to a pressure of 12 bar, we find that
hf = 798.64 kJ/kg; hfg = 1986.2 kJ/kg ; hg = 2784.8 kJ/kg
Enthalpy or total heat of 1 kg of superheated steam,
hsup = hg + cps (tsup − ts)
= 2784.8 + 2 × 67 = 2925.5 kJ/kg
Similarly, from steam tables (Appendix A.1.2), corresponding to a pressure of 0.15 bar
hf = 225.91 kJ/kg; hfg = 2373.1 kJ/kg
Enthalpy or total heat of 1 kg of wet steam,
h = hf + xhfg = 225.91 + 0.95 × 2373.1 = 2480.36 kJ/kg
∴ Drop in enthalpy of the steam = hsup − h = 2925.5 − 2480.36 = 445.14 kJ/kg
Example 2.17
A steam engine obtains steam from a boiler at a pressure of 15 bar and 0.98 dry. It was observed that the steam loses 21 kJ of heat per kg as it flows through the pipe line and pressure remaining constant. Calculate the dryness fraction of the steam at the engine end of the pipeline.
Solution
Given that p = 15 bar, x = 0.98, and heat loss = 21 kJ/kg
From steam table A.1.2 in the Appendix, corresponding to a pressure of 15 bar we find that
hf = 844.87 kJ/kg; hfg = 1947.3 kJ/kg
Enthalpy of wet steam at the boiler end.
h = hf + xhfg = 844.87 + 0.98 × 1947.3 = 2753.22 kJ/kg
Since the steam loses 21 kJ/kg of steam, therefore enthalpy of wet steam at the engine end,
h2 = 2753.22 − 21 = 27332.22 kJ
Let x2 = Dryness fraction of steam at the engine end.
Since the pressure remains constant, therefore hf and hfg is same. We know that
h2 = hf + x2hfg
2732.22 = 844.87 + x2 × 1947.3
or x2 = 0.9692
Example 2.18
A coal fired boiler plant consumes 400 kg of coal per hour. The boiler evaporates 3200 kg of water at 45°C into superheated steam at a pressure of 12 bar and 274.5°C. If the calorific value of the fuel is 32600 kJ/kg of coal, then determine the following:
- Equivalent evaporation
- Efficiency of boiler
Assume specific heat of superheated steam as 2.1 kJ/kg.K.
Solution
Given that mf = 400 kg/h, ms = 3200 kg/h, feed water temperature, t1 = 45°C, p = 12 bar,
Tsup = 274.5°C, C.V. = 32600 kJ/kg, cps = 2.1 kJ/kg.K
From steam table A.1.2 in the Appendix, at 12 bar, we have
Ts = 187.99°C, hg = 2784.2 kJ/kg
From steam table A.1.3 in the Appendix, using linear interpolation, enthalpy of superheated steam at exit of boiler (12 bar and 274.5°C) is found to be
hsup = 2989.29 kJ/kg
From steam table A.1.1 in the Appendix, enthalpy of water at 45°C,
hf1 = 188.42 kJ/kg
- Equivalent evaporation:
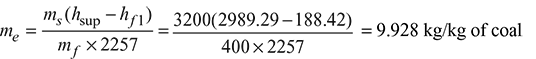
- Boiler efficiency:
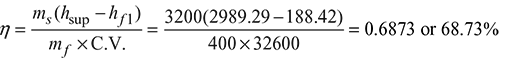
Example 2.19
During a trial on an oil-fired smoke tube boiler for 1 h, the following data were recorded:
Steam pressure, p = 14 bar
Amount of water evaporated, m1= 5400 kg
Condition of steam, x = 0.92 dryness fraction
Amount of fuel burnt, mf = 540 kg
Calorific value of fuel used, C.V. = 42000 kJ/kg
Temperature of steam leaving the superheater, Tsup = 250°C
Temperature of feed water, t1 = 50°C
Determine the equivalent evaporation from and at 100°C with and without superheater, boiler efficiency, and the percentage of heat utilised in the superheater.
Solution
Given that p = 14 bar, m1 = 5400 kg, x = 0.92, mf = 540 kg, C.V. = 42000 kJ/kg, Tsup = 250°C, and t1 = 50°C
Amount of steam generated per kg of fuel,
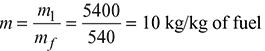
Enthalpy of wet steam at 15 bar, 0.92 dry,
h = hf + x.hfg = 845.2 + (0.92 × 1947.2) = 2636.6 kJ/kg
From steam table A.1.2 in the Appendix, enthalpy of wet steam at 14 bar, 0.92 dry,
h = hf + x.hfg = 830.29 + (0.92 × 1959.7) = 2633.2 kJ/kg
From steam table A.1.3 in the Appendix, Enthalpy of superheated steam at 14 bar, 250°C,
hsup = 2927.2 kJ/kg
Equivalent evaporation without superheated from and at 100°C.
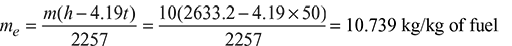
Equivalent evaporation with superheater from and at 100°C.
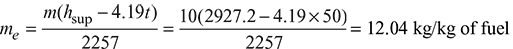
Boiler efficiency, ![]()

Percentage of heat utilized in the superheater,
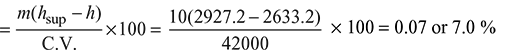
Example 2.20
Following particulars refer to a steam power plant consisting of a boiler, superheater, and an economiser:
Steam pressure, p = 20 bar
Mass of steam generated, m1 = 10000 kg/h
Mass of coal used, mf = 1300 kg/h
Calorific value of coal, C.V. = 29000 kJ/kg
Temperature of feed water entering the economiser, t1 = 35°C
Temperature of feed water leaving the economiser, t2 = 105°C
Temperature of superheated steam leaving the superheater, tsup = 350°C
Dryness fraction of steam leaving the boiler, x = 0.98
Determine the following:
- Overall efficiency of the plant
- Equivalent evaporation from and at 100°C
- The percentage of heat utilised in boiler, superheater, and economiser
Solution
Given that p = 20 bar, m1 = 10000 kg/h, mf = 1300 kg/h, C.V. = 29000 kJ/kg, t1 = 35°C, t2 = 105°C, tsup = 350°C, x = 0.98
From steam table A.1.1 in the Appendix, enthalpy of feed water entering the economiser,
hf1 = 146.66 kJ/kg
Enthalpy of feed water leaving the economiser at 105°C,
hf2 = 440.13 kJ/kg
From steam table A.1.2 in the Appendix, enthalpy of steam leaving the boiler,
h = hf + x.hfg = 908.77 + 0.98 × 1890.7 (at 20 bar, 0.98 dry)
= 2761.66 kJ/kg
From steam table A.1.3 in the Appendix, enthalpy of steam leaving the superheater,
hsup = 3137.0 kJ/kg (at 20 bar, 350°C)
Mass of steam generated per kg of fuel,

- Overall efficiency of the plant:
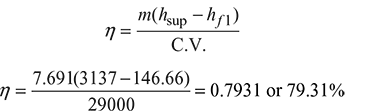
- Equivalent evaporation from and at 100°C:
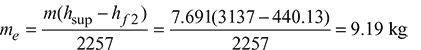
- Percentage of heat utilised in the boiler:Heat supplied in 1 kg of fuel = 29000 kJ
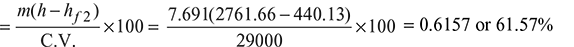 Percentage of heat utilised in superheater:
Percentage of heat utilised in superheater: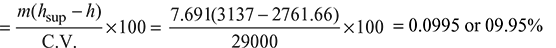 Percentage of heat utilised in economiser:
Percentage of heat utilised in economiser: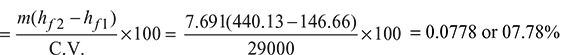
Note: Superheater is considered as a part of the boiler for calculation of equivalent evaporation.
Example 2.21
The following readings were recorded during a boiler trial of 6 h duration:
Mean steam pressure = 12 bar
Mass of steam generated = 40000 kg
Mean dryness fraction of steam = 0.85
Mean feed water temperature = 30°C
Coal used = 4000 kg
Calorific value of coal = 33400 kJ/kg
Calculate the following:
- Factor of equivalent evaporation
- Heat rate of boiler in kJ/hr
- Equivalent evaporation from and at 100°C
- Efficiency of the boiler
Solution
Given that p = 12 bar, m = 40000 kg, x = 0.85, t1 = 30°C, mf = 4000 kg, C.V. = 33400 kJ/kg, and duration of trial, T = 6 h
From steam table A.1.2 in the Appendix at 12 barhf = 798.64 kJ/kg, hfg = 1986.2 kJ/kg
∴ Enthalpy of steam, h = hf + x.hfg = 798.64 + 0.85 × 1986.2 = 2486.91 kJ/kg
From steam table A.1.1 in the Appendix, enthalpy of feed water at 30°C, hf 1 = 125.77 kJ/kg
- Factor of equivalent evaporation:
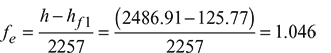
- Heat rate of boiler:
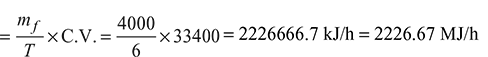
- Equivalent evaporation from and at 100°C:
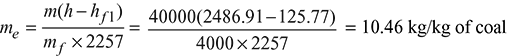
- Boiler efficiency:
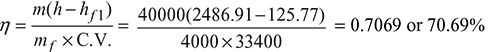
Example 2.22
During a trial for 8 h on a boiler, the following data were obtained:
Pressure of steam leaving the boiler = 14 bar
Condition of steam = 0.97 dryness fraction
Steam produced = 26700 kg
Temperature of feed water = 50°C
Mass of coal fired = 4260 kg
Calorific value of coal = 28900 kJ/kg
Air fuel ratio = 17
Temperature of flue gases leaving the boiler = 344°C
Boiler house temperature = 21°C
Specific heat of flue gases = 1.1 kJ/kg.K
Determine the following:
- Boiler efficiency
- Equivalent evaporation
- Heat lost to flue gases expressed as percentage of heat supplied
Solution
Given that p = 14 bar, x = 0.97, m = 26700 kg, t1 = 50°C, mf = 4260 kg, C.V. = 28900 kJ/kg, air−fuel ratio, ![]() = 17, tf = 344°C, t0 = 21°C, Cpg = 1.1 kJ/kg.K, and duration of boiler trial, T = 8 h
= 17, tf = 344°C, t0 = 21°C, Cpg = 1.1 kJ/kg.K, and duration of boiler trial, T = 8 h
- From steam table A.1.2 in the Appendix, at 14 bar, hf = 830.29 kJ/kg and hfg = 1959.7 kJ/kgEnthalpy of steam, h = (hf + x.hfg) at 14 bar = 830.29 + 0.97 × 1959.7 = 2731.2 kJ/kgFrom steam table A.1.1 in the Appendix, enthalpy of water at 50°C, hf 1 = 209.31 kJ/kg∴ Efficiency of boiler,
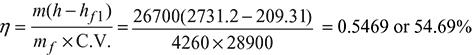
- Equivalent evaporation:
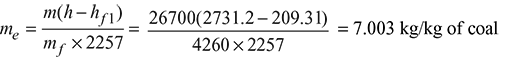
- Fuel burnt/hour,
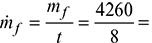 532.5 kg/hHeat supplied in fuel = ṁf × C.V. = 532.5 × 28900 = 15389250.0 kJ/hMass of flue gases formed/hour,
532.5 kg/hHeat supplied in fuel = ṁf × C.V. = 532.5 × 28900 = 15389250.0 kJ/hMass of flue gases formed/hour,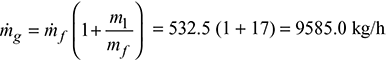 Heat carried away by flue gases,= ṁg × cpg (tf − t0) = 9585 × 1.1 (344 − 21) = 3405550.5 kJ/hHeat loss of flue gases as per cent of heat supplied
Heat carried away by flue gases,= ṁg × cpg (tf − t0) = 9585 × 1.1 (344 − 21) = 3405550.5 kJ/hHeat loss of flue gases as per cent of heat supplied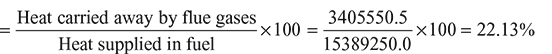
Example 2.23
A gas-fired boiler operates at a pressure of 100 bar. The feed water temperature is 255°C. Steam is produced with a dryness fraction of 0.9, and in this condition, it enters a superheater. Superheated steam leaves the superheater at a temperature of 450°C. The boiler generates 1200 tonne of steam per hour with an efficiency of 92%. The gas used has a calorific value of 38 mJ/m3. Determine the following:
- The heat transfer per hour in producing wet steam in the boiler
- The heat transfer per hour in producing superheated steam in the superheater
- The gas used in m3/hour
Solution
Given that p = 100 bar, t1 = 256°C, x = 0.9, tsup = 450°C, m = 1200 tonne = 1200 × 103 kg
η = 0.92, C.V. = 38 MJ/m3 = 38 × 103 kJ/m3
- From table A.1.1 in the Appendix, enthalpy of feed water: hf 1 = 1109.72 kJ/kgFrom table A.1.2 in the Appendix, enthalpy of steam at 100 bar, 0.9 dryh = hf + x.hfg = 1407.53 + 0.9 × 1317.1 = 2592.92 kJ/kgFrom table A.1.3 in the Appendix, Enthalpy of superheated steam at 100 bar, 450°Chsup = 3240.8 kJ/kg∴ Heat transfer/h producing wet steam in boiler= m(h − hf1) = 1200 × 103 (2592.92 − 1109.72)= 17.7984 × 108 kJ/h
- Heat transfer/hour in producing superheated steam in superheater:= m(hsup − h) = 1200 × 103 (3240.8 − 2592.92)= 77.7456 × 107 kJ/h
- Let the gas used in m3/hour be Vf.
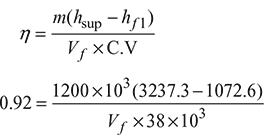 Vf = 73149.2 m3/h
Vf = 73149.2 m3/h
Leave a Reply