In Mollier diagram (Fig. 2.3), the vertical ordinate represents the enthalpy and the horizontal ordinate represents the entropy. The regions above and below the saturation line represent the superheated and wet conditions of steam, respectively. The lines of constant dryness fraction are shown in the wet steam region, whereas the lines of constant temperature and pressure in the region of both wet and superheat are shown.
From the thermodynamic property relationship, one can write,
Tds = dh − vdp
For constant pressure process, we have
Tds = dh
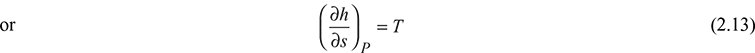
From Eq. (2.13), it is clear that the slope of the constant pressure lines is equal to the corresponding saturation temperatures. It may be noted that as the pressure increases, the saturation temperature also increases. This is the reason for which constant pressure lines (isobar) are divergent on the h−s diagram. The saturated liquid line and saturated vapour line meet at critical point.
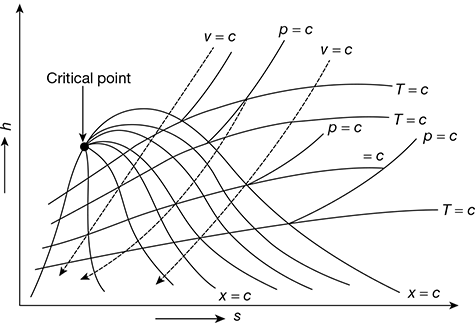
Figure 2.3 Mollier diagram for steam
VARIOUS PROCESSES FOR STEAM
1 Constant Volume Process
The constant volume heating process is shown on the p−v and T−s diagrams in Fig. 2.4. Consider 1 kg of wet steam at a certain pressure p1, and dryness fraction x1 is heated to state 2 in the wet region and then to state 3 in the superheated region.
Initial volume of steam v1 = vf 1 + x1(vg1 − vf1)
Final volume of steam in wet region v2 = vf2 + x2(vg2 − vf2)
Since the volume remains constant,
vf1 + x1(vg1 − vf1) = vf2 + x2(vg2 − vf2)
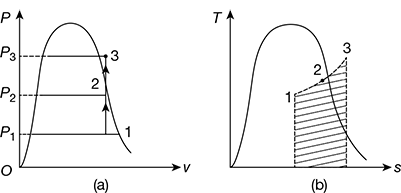
Figure 2.4 Constant volume heating: (a) p−v diagram, (b) T−s diagram
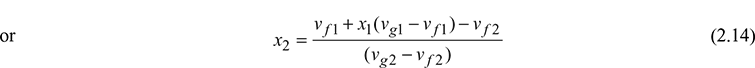
From steam table, internal energy of steam can be found as follows:
Initial internal energy of steam, u1 = uf1 + x1 ufg1
Final internal energy of steam in the wet region, u2 = uf2 + x2 ufg2
Since dv = 0, therefore δW = 0

For constant volume heating process, v2 = v3. After knowing the value of v3, the condition of steam can be found from superheated steam table. If pressure of state 3 is given (=p3), then from known values of p3 and v3, temperature of state 3 can be obtained. Similarly, if temperature at state 3 (=T3) is given, pressure can be found from known values of T3 and v3.
2 Constant Pressure Process
The constant pressure heating process is shown in Fig. 2.5. In this case, p1 = p2 = p (say).
For wet steam, during heating process 1–2,
Work done per kg of steam, w = pdv = p(v2 − v1)
where v1 = vf + x1(vg − vf) and v2 = vf + x2(vg − vf)
The specific enthalpy of steam, h1 = hf + x1 hfg and h2 = hf + x2 hfg
From first law of thermodynamics, heat transfer per kg of steam during constant pressure heating
q = h2 − h1
q = (hf + x2hfg) − (hf + x1hfg)

For superheated steam, specific volume and specific enthalpy of steam can be found from the superheated steam table. Then, during heating process 2-3,
Work done per kg of steam, w = pdv = p(v3 − v2) = p(vsup − v2)
and heat transfer per kg of steam

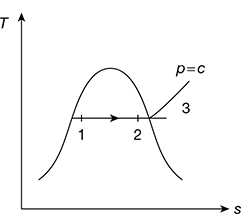
Figure 2.5 Constant pressure expansion
3 Isothermal Process
The isothermal heating process is shown in Fig. 2.6. The isothermal process for wet steam will be same as the constant pressure process. However, in the superheated region, the steam will behave as a gas and shall follow Boyle’s law, pv = const. Therefore, the process will be hyperbolic.
In the wet region,
q = h2 − h1 and w = p1 (v2 − v1)
In the superheated region,
q = h3 − h2 and w = p1 (v3 − v2)
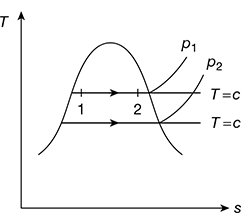
Figure 2.6 Isothermal process
4 Hyperbolic Process
During a hyperbolic process, pv = const. If the superheated vapour behaves as an ideal gas, then the process during which pv = const. may be regarded isothermal, as shown in Fig. 2.7.
For 1 kg of steam in the wet region,
Initial volume of steam v1 = vf1 + x1(vg1 − vf1)
Final volume of steam v2 = vf2 + x2(vg2 − vf2)
As pv = const., therefore

or 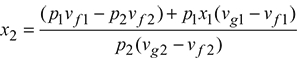
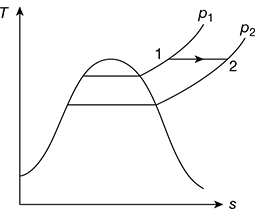
Figure 2.7 Hyperbolic expansion
Now, h1 = hf 1 + x1hfg1 and h2 = hf 2 + x2hfg2 for wet steam
and hs2 = hg2 + cp (Tsup − T2) for superheated steam
Change is internal energy,


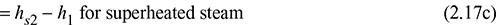

where v1 and v2 are the specific volumes of steam before and after heating.
Total heat supplied, q = w + (u2 − u1)
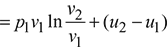

5 Reversible Adiabatic or Isentropic Process
The isentropic process of expansion (1-2) is shown in Fig. 2.8 on T−s diagram. The initial condition of steam may be wet, dry, or superheated. For initially wet steam as shown in Fig. 2.8(a), we have
s1 = s f1 + x1s fg1
s2 = s f2 + x2s fg2
Since s1 = s2, therefore
s f 1 + x1s fg1 = s f 2 + x2s fg2

For initially dry steam (Fig. 2.8(b)), s1 = sg1
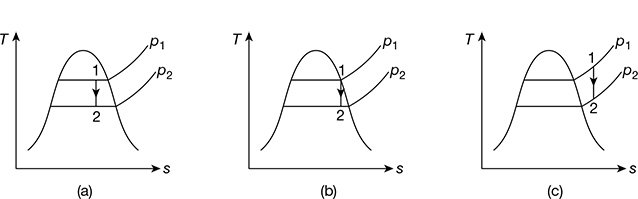
Figure 2.8 Isentropic expansion: (a) Expanded steam wet, (b) Expanded steam dry saturated, (c) Expanded steam superheated
Then, sg1 = s f2 + x2s fg2

For initially superheated steam at temperature Tsup, s1 can be calculated from superheated steam table. After expansion, state 2 may be superheated (Fig. 2.8(c)), dry or wet. If the state after expansion is superheated, then the condition of steam can be calculated from the relationship s1 = s2. Note that if pressure is specified, then temperature can be calculated or otherwise if temperature is specified, then pressure can be calculated.
Now q = w + (u2 − u1)
where u1 = u f1 + x1u fg1 for wet steam
= ug1 for dry steam
= usup for superheated steam
Similarly, u2 can be found depending on the condition of steam after expansion.
But q = 0
w = u1 − u2
6 Polytropic Process
Let the steam expand from condition 1 to 2 according to the law pvn = constant. Then for wet steam,
Initial volume of steam, v1 = vf1 + x1(vg1 − vf1)
Final volume of steam v2 = vf2 + x2(vg2 − vf2 )
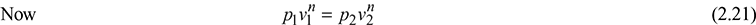
or
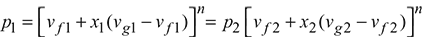
or 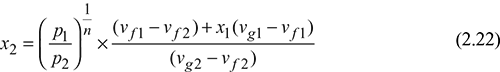
Also u1 = uf1 + x1ufg1
u2 = uf2 + x2ufg2

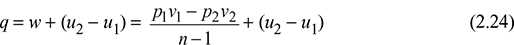
n = 1.13 for wet steam and 1.3 for superheated steam
For superheated steam on heating from wet steam, v2 can be calculated from superheated steam table for the known pressure and temperature conditions. Similarly, the condition of steam can also be found after knowing the value of v2 from Eq. (2.21).
7 Throttling Process
The throttling process occurs when steam is expanded through a small aperture as in the case of throat of a nozzle. During this process, no work is done and the enthalpy remains constant. The throttling process is shown on T−s and h−s diagrams in Fig. 2.9.
h1 = hf1 + x1hfg1
h2 = hf2 + x2hfg2 for wet steam after expansion
= hf2 + hfg2 + cps (Tsup − Ts), for superheated steam after expansion
Now h1 = h2
For wet steam after expansion

For superheated steam after expansion

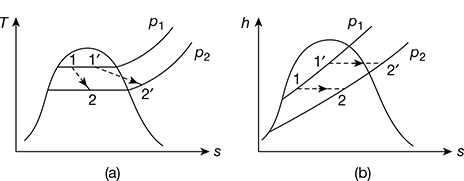
Figure 2.9 Throttling process: (a) T−s diagram, (b) h−s diagram
Leave a Reply