Consider 1 kg of ice under atmospheric pressure and at a temperature of −10°C contained in a vessel. If we heat the ice gradually keeping the pressure constant, the following changes will take place, as shown in Fig. 2.1:
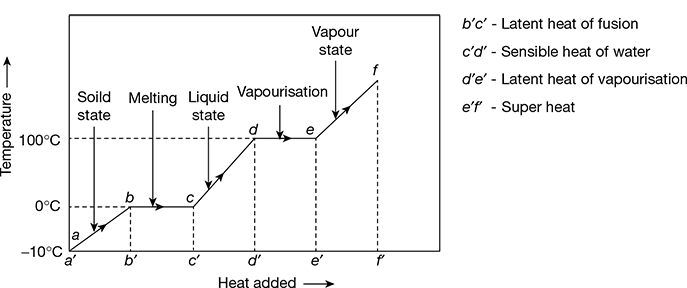
Figure 2.1 Constant pressure temperature-heat added diagram for steam and water
- The temperature of ice will gradually increase till it just reaches the freezing temperature (represented by point ‘b’), which is 0°C at atmospheric pressure. This is shown by the straight line ab.
- On addition of more heat at point b, the ice starts melting, and with no rise in temperature till the whole ice has melted, up to point c, the ice is converted into water. The heat added during the process bc is called the latent heat of fusion of ice or simply latent heat of ice.
- Gradual addition of more heat beyond the point c leads to gradual increase in temperature till the water reaches the vapourisation or boiling point ‘d’. Under atmospheric pressure of 1.01325 bar, it is 100°C.
- On further heating beyond point d, the water will gradually turn into steam, with no rise in temperature. This steam contains some water particles and is called the wet steam. With further addition of heat, all the water particles in the steam disappear and this steam is called dry saturated steam or simply dry steam. This corresponds to point ‘e’. The heat added during de is called the latent heat of vapourisation of steam or simply latent heat of steam.
- If the dry saturated steam is further heated beyond point e, keeping the pressure constant, the process is called superheating. The steam so obtained is called the superheated steam. Let the steam be superheated to temperature ts°C, represented by point ‘f’. Then (ts − t)°C is called the degree of superheat. The heat added during process ef is called the heat of superheat.
Leave a Reply