It is used to determine the higher calorific value of gaseous fuels. Figure 1.4 shows the components of the calorimeter. It consists of a gas burner B with arrangements to measure flow quantity and pressure of gas supplied to burner. A gas pressure regulator is used in the gas supply line to control gas flow and pressure fluctuation. The fuel burns within a cylindrical container surrounded by a cooling coil. Cooling water is supplied to the cooling coil from a constant heat tank. The outer casing of the calorimeter is provided with heavy insulation to prevent any heat loss to the surroundings.
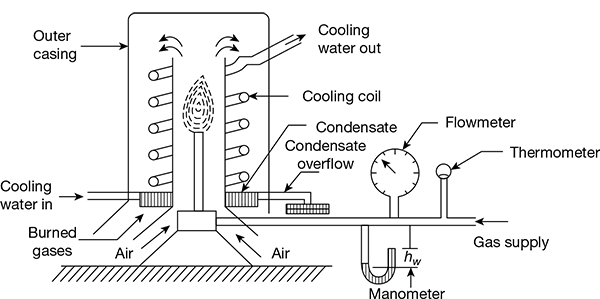
Figure 1.4 Boys gas calorimeter
1.20.2 Working
The flue gas moves up the cylindrical container and flows down from the top of the container. The steam formed due to the combustion of hydrogen in the fuel and carried with the gases condenses around the cooling coil and drips down into the trap below. The overflow of the condensate is taken out in a glass beaker.
Calculations
Let Vg′ = volume of gas consumed, m3
mw = mass of water circulated, kg
(∆θ)w = rise in temperature of water circulated, °C
hw = pressure of gas above atmosphere, cm of water
hb = barometer reading, cm of Hg
θg = temperature of gas supply, °C
Volume of gas at normal temperature and pressure (NTP),
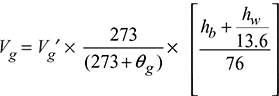
The calorific value of fuel is given by
Vg × HCV = mw × (Δθ)w
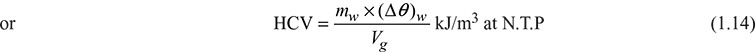
Example 1.1
A coal has the following composition by mass:
C = 0.89, H2 = 0.03, S = 0.01, O2 = 0.02, and N2 = 0.03
and remaining is ash. Calculate the HCV and LCV of coal.
HCV = 35000 C + 143000 ![]() + 9160 S
+ 9160 S
= 35000 × 0.89 + 143000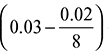 + 9160 × 0.01
+ 9160 × 0.01
= 35174 kJ/kg
LCV = HCV – 9H × 2460 = 35174 – 9 × 0.03 × 2460
= 34510 kJ/kg
Example 1.2
The following observations were made to find the HCV and LCV of a solid fuel by using a bomb calorimeter:
| Mass of fuel in crucible, mf | = 0.8 gram |
| Mass of fuse wire, mfw | = 0.02 gram |
| CV of fuse wire | = 6500 kJ/kg |
| Mass of water in calorimeter, mw | = 1.90 kg |
| Water equivalent of calorimeter, mc | = 0.35 kg |
| Observed temperature rise, ∆θ | = 2.97°C |
| Cooling correction | = 0.03°C |
| The fuel contains 90% C and 4% H2, the rest being ash. | |
Solution
Corrected temperature rise, ∆θc = 2.97 + 0.03 = 3°C
mf × HCV + mfw × C.V. = cpw (mw + mc). Δθc
or 0.8 × 10–3 × HCV + 0.02 × 10–3 × 6500 = 4.2 (1.90 + 0.35) × 3
or HCV = 35275 kJ/kg
LCV = HCV – 9H × 2460 = 35275 – 9 × 0.04 × 2460
= 34389.4 kJ/kg
Example 1.3
The following observations were made by using a Boys gas calorimeter to determine the LCV of a gaseous fuel:
Gas burned = 50 litres
Gas pressure = 4.5 cm of water above atmospheric pressure
Barometer reading = 750 mm of Hg
Temperature of gas = 30°C
Water circulated through the calorimeter = 19 kg
Rise in temperature of water = 10°C
Condensate collected during the test = 60 gram.
Determine the HCV and LCV of the gas at 0°C and 760 mm of mercury pressure.
Solution
Volume at N.T.P., Vg = Vg′
= 50 × 10–3 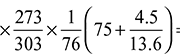 0.04465 m3
0.04465 m3
HCV = 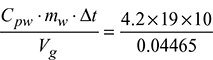 = 17872 kJ/m3 at N.T.P.
= 17872 kJ/m3 at N.T.P.
Condensate formed per m3 of gas used at N.T.P.
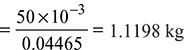
LCV = HCV –1.1198 × 2460 = 15117 kJ/m3
Example 1.4
The percentage composition by mass of a solid fuel is as follows: C = 90%, H2 = 3.5%, O2 = 3%, N2 = 1%, S = 1%, and the remainder being ash. Find (a) the mass of air required per kg of fuel of complete combustion, (b) the mass analysis of the dry products of combustion, (c) If 60% excess air is supplied in actual combustion, determine the mass of flue gases per kg of fuel, and (d) Volumetric analysis of the products per kg of fuel.
Solution
-
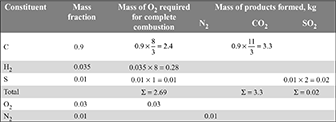 Total O2 required for complete combustion = 2.69 – 0.03 = 2.66 kg/kg of fuelAir required = 2.66 ×
Total O2 required for complete combustion = 2.69 – 0.03 = 2.66 kg/kg of fuelAir required = 2.66 × 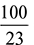 = 11.58 kg/kg of fuel
= 11.58 kg/kg of fuel - Mass analysis of dry products of combustion: N2 = 11.58 × 0.77 + 0.01 = 8.92 kg
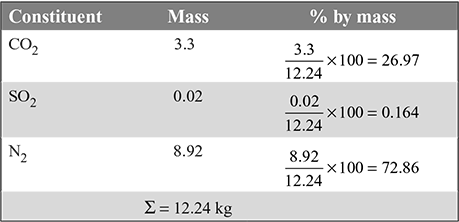
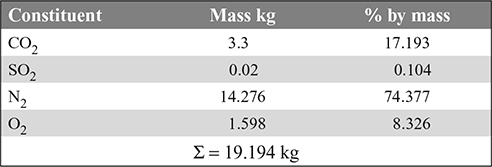
- For 60% excess air,N2 = 11.58 × 1.6 × 0.77 + 0.01 = 14.276 kgO2 = 11.58 × 0.6 × 0.23 = 1.598 kg
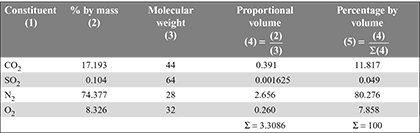
- Volumetric analysis
Example 1.5
The ultimate analysis of a coal is as follows:
C = 82%, H2 = 5%, O2 = 8%, and the rest is ash.
The volumetric analysis of dry flue gases is:
CO2 = 10%, CO = 2%, O2 = 8%, and N2 = 80%.
Find (a) air supplied per kg of fuel, (b) flue gases formed per kg of fuel, (c) percentage of excess air supplied, (d) heat carried away by the products of combustion per kg of fuel if exit temperature of flue gases is 380°C, and (e) heat lost by incomplete combustion. Assume C.V. of carbon = 35000 kJ/kg when it burns to CO2 and 10200 kJ/kg when it burns to CO. Air contains 23% of oxygen by mass. cp for dry flue gas = 1 kJ/kgK and for steam, cp = 2.10 kJ/kgK.
Solution
- Mass of carbon per kg flue gas = mass of C from CO2 + mass of C from CO
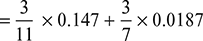 = 0.0481 kg/kg of flue gasCarbon in 1 kg of fuel = 0.82 kgMass of dry flue gases formed per kg of fuel, mg =
= 0.0481 kg/kg of flue gasCarbon in 1 kg of fuel = 0.82 kgMass of dry flue gases formed per kg of fuel, mg = 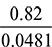 = 17.048 kgWater formed by 0.05 kg of H2 per kg of fuel = 0.05 × 9 × 0.45 kgMass of combustible matter per kg of fuel + air supplied per kg of fuel = mass of products0.82 + 0.05 + 0.08 + ma = 17.048 + 0.45or ma =
= 17.048 kgWater formed by 0.05 kg of H2 per kg of fuel = 0.05 × 9 × 0.45 kgMass of combustible matter per kg of fuel + air supplied per kg of fuel = mass of products0.82 + 0.05 + 0.08 + ma = 17.048 + 0.45or ma = 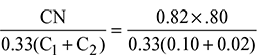 = 16.55/kg fuel
= 16.55/kg fuel -
- Theoretical air required for complete combustion of fuel
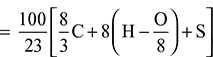
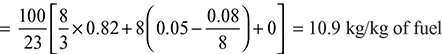 Excess air supplied = 16.55 – 10.9 = 5.65 kg of fuelPercentage of excess air =
Excess air supplied = 16.55 – 10.9 = 5.65 kg of fuelPercentage of excess air = 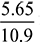 × 100 = 51.83%
× 100 = 51.83% - Heat carried by flue gasses = Heat carried by dry flue gases + Heat carried by steam formed= (1 × 17.048 × 380) + [2460 + 2.1 × (380 –15) × 0.5183]= 9335.5 kJ/kg of fuel burned
- Carbon in fuel burned to CO =
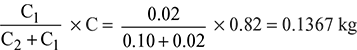 The burning of carbon to CO instead of CO2 causes a loss of (35000 – 10200)= 24800 kJ/kg of C burned to COHeat loss = 24800 × 0.1367 = 3389.3 kJ/kg of fuel
The burning of carbon to CO instead of CO2 causes a loss of (35000 – 10200)= 24800 kJ/kg of C burned to COHeat loss = 24800 × 0.1367 = 3389.3 kJ/kg of fuel
Example 1.6
The flue gas analysis by volume for burning of C6H14 is as follows:
CO2 = 8.5%, CO = 7.8%, and N2 = 83.7%
Calculate (a) the air-fuel ratio required for chemically correct combustion, and (b) the air-fuel (A:F) ratio as a percentage of the chemically correct mixture A:F ratio.
Solution
2C6H14 + 19O2 = 12CO2 + 14H2O
2 × 86 + 19 × 32 = 12 × 44 + 14 × 18
For complete combustion of 1 kg of fuel, oxygen required
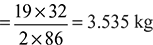

∴ A:F ratio for complete combustion = 15.37: 1
The percentage of carbon in the fuel = ![]() = 83.72%
= 83.72%
Mass of air supplied per kg of fuel burned,
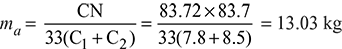
∴ A:F ratio of the mixture supplied = 13.03:1
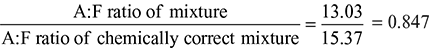
Example 1.7
The volumetric analysis of a sample of producer gas is as follows:
H2 = 20%, CH4 = 3%, CO = 22%, CO2 = 8%, and N2 = 47%.
- Find the volume of air required for complete combustion of 1 m3 of the gas.
- If 50% excess air is supplied, find the percentage contraction in volume after the products of combustion have been cooled.
Solution
- Minimum volume of air required for complete combustion of 1 m3 of fuel
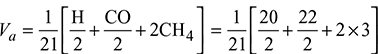 = 1.29 m3/m3 of gas
= 1.29 m3/m3 of gas - The constituents formed with 50% excess air are:CO2 = 0.08 + CO2 formed from CO + CO2 formed from CH4= 0.08 + 0.22 × 1 + 0.03 × 1 = 0.33 m3N2 = 0.47 + N2 from the air supplied= 0.47 + 1.29 × 1.5 × 0.79 = 2 m3O2 = O2 from the excess air only= (1.29 × 0.5) × 0.21 = 0.135 m3Total dry flue gases formed = CO2 + N2 + O2 = 0.33 + 2.00 + 0.135 = 2.465 m3Total volume before combustion = volume of gas + volume of air supplied= 1 + 1.29 × 1.5 = 2.935 m3Percentage contraction in volume after combustion and cooling
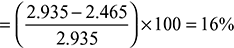
Example 1.8
A sample of coal has the following percentage composition by weights:
C = 82%, H2 = 10%, O2 = 6%, N2 = 1%, and S = 1%.
Calculate the minimum quantity of air required, for the complete combustion of 1 kg of coal. If 20% excess air is used in the combustion, what is the air-fuel ratio?
Solution
Minimum air per kg of fuel
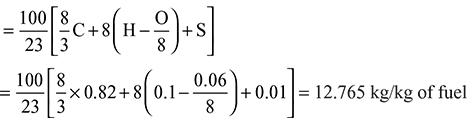
With 20% excess air,
A:F = 12.765 × 1.2 = 15.318 kg/kg of fuel
Example 1.9
A fuel gas has composition by volume: H2 = 50%, CO = 40%, CO2 = 6% and N2 = 4%. (a) Calculate the volume of air required for complete combustion of 1 m3 of the gas, (b) If the dry exhaust gas from an engine using this fuel contains 9.2% by volume of CO2 and no CO, calculate the air to gas ratio which has been used. Calculate the percentage of O2 by volume in the dry exhaust.
Solution
- For complete combustion of fuel gas, the chemical equation is:
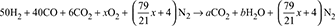 where
where  oxygen – fuel ratio by volume.Comparing coefficients of H2, C and O2 on both sides, we getb = 50, a = 40 + 6 = 46
oxygen – fuel ratio by volume.Comparing coefficients of H2, C and O2 on both sides, we getb = 50, a = 40 + 6 = 46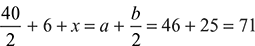 x = 45 m3 of O2 per 100 m3 of fuel gas.A:F =
x = 45 m3 of O2 per 100 m3 of fuel gas.A:F = 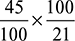 = 2.14 m3 of air/m3 of gas.
= 2.14 m3 of air/m3 of gas. 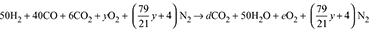 where
where 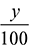 = new oxygen – fuel ratio by volumeC balance: d = 40 + 6 = 46CO2 balance: 0.092 =
= new oxygen – fuel ratio by volumeC balance: d = 40 + 6 = 46CO2 balance: 0.092 = 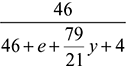 O2 balance:
O2 balance: 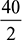 + 6 + y = 46 + eSolving for y, we get y = 104 and e = 59Air to gas ratio by volume =
+ 6 + y = 46 + eSolving for y, we get y = 104 and e = 59Air to gas ratio by volume = 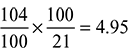 % volume of O2 in dry gas =
% volume of O2 in dry gas = 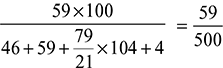 × 100 = 11.8%
× 100 = 11.8%
Example 1.10
The composition of a hydrocarbon fuel by mass is: C = 85%, H2 = 13%, and O2 = 2%. Calculate (a) the chemically correct mass of air required for the complete combustion of 1 kg of fuel, (b) the volumetric and gravimetric analysis of the products of combustion, and (c) dew point temperature if the total pressure is 1 bar and (d) If the products of combustion are cooled to 15°C, calculate the mass of water vapour condensed.
Solution
C + O2 → CO2
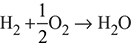
Moles of O2 required = ![]() = 0.103958 moles of O2/kg of fuel
= 0.103958 moles of O2/kg of fuel
- Mass of air required = 0.103958 ×
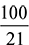 × 29 = 14.356 kg of air/kg fuel
× 29 = 14.356 kg of air/kg fuel - The volumetric and gravimetric analysis of products of combustion:
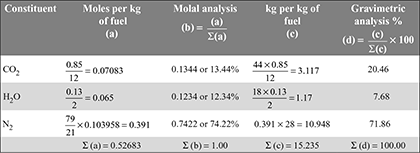
- Partial pressure of H2O,
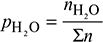 × p = 0.1234 × 1 = 0.1234 barDew point is the saturation temperature corresponding to 0.1234 bar. From steam tables,tdp = 49.97°C.
× p = 0.1234 × 1 = 0.1234 barDew point is the saturation temperature corresponding to 0.1234 bar. From steam tables,tdp = 49.97°C. - Temperature of products of combustion on cooling (15°C) is less than tdp. Hence, some water vapour will condense. The vapour pressure will then be equal to the saturation vapour pressure corresponding to 15°C.From steam tables, ps = 0.017051 bar and vg = 77.926 m3/kg.The volume occupied by the water vapour is equal to the volume occupied by CO2 and N2 at their partial pressure of (1 – 0.017051) = 0.982949 bar.Volume,
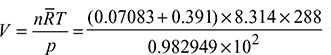 = 11.25 m3Mass of vapour in combustion products =
= 11.25 m3Mass of vapour in combustion products = 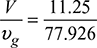 = 0.14437 kgMass of condensed vapour = 1.17 – 0.14437 = 1.0256 kg
= 0.14437 kgMass of condensed vapour = 1.17 – 0.14437 = 1.0256 kg
Example 1.11
Octane, C8H18’ is burned with dry air. The molar analysis of the dry products is CO2 = 10%; CO = 0.4%; O2 = 3%, and N2 = 86.6%. Determine the air fuel ratio on a mass basis and the dew point temperature of the products if the products are at 1 atm pressure. Dry air contains 3.76 moles of nitrogen per mole of oxygen.
[IES, 1994]
Solution
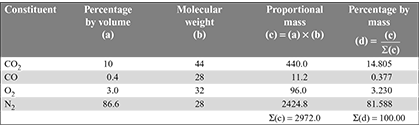
Mass of C per kg of dry flue gas = Mass of C from CO2 + Mass of C from CO
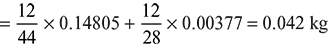
Percentage of C in fuel = ![]() × 100 = 84.21%
× 100 = 84.21%
Mass of dry flue gases formed per kg of fuel = ![]() = 20.05 kg
= 20.05 kg
Mass of air supplied per kg of fuel = 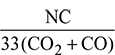
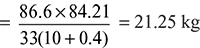
Air/Fuel ratio = 21.25:1.
The dew point temperature of products is the saturation temperature at the partial pressure of water vapour.
C8H18 + 12.5O2 + 12.5 (3.76) N2 → 8CO2 + 9H2O + 47N2
12.5 moles of O2 are required to burn 1 mole C8H18 and 8 moles of H2O are produced.
Stoichiometric mole fraction of water vapour (xH2O) formed,

pH2O = xH2O atm = 0.1636 bar
The saturation temperature of water vapour corresponding to their partial pressure of 0.1636 bar is, ts = 54°C.
Example 1.12
An oil fired boiler uses a fuel which contains 84% carbon and 16% hydrogen by weight. The volumetric analysis of the dry flue gases was found to be CO2 = 9%, CO = 2%, O2 = 5%, and N2 = 84%. Assuming complete combustion of the hydrogen in the fuel, determine the quantity of carbon remaining unburnt in each kg of the fuel fired in the furnace. Determine the actual air-fuel ratio and the excess air factor.
[IES, 1983]
Solution
C + O2 = CO2
12 + 32 = 44
0.84 kg of C requires ![]() × 0.84 = 2.24 kg of O2
× 0.84 = 2.24 kg of O2
2H2 + O2 = 2H2O
4 + 32 = 36
0.16 kg of H2 requires ![]() × 0.16 = 1.28 kg of O2
× 0.16 = 1.28 kg of O2
Total O2 required for complete combustion of 1 kg of fuel = 2.24 + 1.28 = 3.52 kg
Amount of air required for complete combustion of 1 kg of fuel

Air/fuel ratio = 15.3:1
Mass analysis of dry flue gases:
Mass of C per kg of dry flue gas
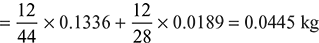
Mass of C per kg of fuel = 0.84 kg
Mass of dry flue gases per kg of fuel = ![]() = 18.876 kg
= 18.876 kg
Mass of N2 per kg of fuel = 18.876 × 0.7935 = 14.978 kg
Mass of air required per kg of fuel = ![]() = 19.45 kg
= 19.45 kg
Excess air supplied = 19.45 – 15.3 = 4.15 kg
Excess air factor = ![]() = 0.2714 or 27.14%
= 0.2714 or 27.14%
Example 1.13
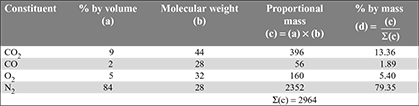
The composition of coal burnt during a boiler trial was as follows:
C = 62%, H2 = 3.6%, O2 = 7.4%, and ash = 27%.
The volumetric analysis of the fuel gas showed the following:
CO2 = 10.25%, CO = 0.80%, O2 = 8.54%, and N2 = 80.41%.
Determine
- the air-fuel ratio
- the percentage of excess air, and
- the mass of CO2, CO, O2, and N2 in the flue gases per kg of coal burnt.[IES, 1986]
Solution
Conversion of volumetric analysis to mass analysis.
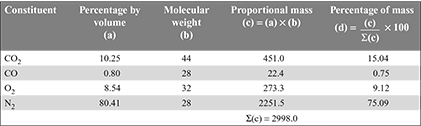
- Mass of carbon per kg of dry flue gas= mass of C in CO2 + mass of C in CO
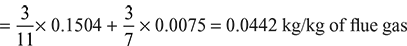 Mass of dry flue gases formed per kg of fuel
Mass of dry flue gases formed per kg of fuel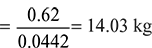 Mass of steam formed per kg of fuel = 0.036 × 9 = 0.324 kgMass of combustible matter + air supplied per kg of fuel = mass of products C + H2 + O2 + air supplied = mass of products0.62 + 0.036 + 0.074 + ma = 14.03 + 0.324or ma = 13.624 kg/kg of fuelAir/fuel ratio = 13.624:1.
Mass of steam formed per kg of fuel = 0.036 × 9 = 0.324 kgMass of combustible matter + air supplied per kg of fuel = mass of products C + H2 + O2 + air supplied = mass of products0.62 + 0.036 + 0.074 + ma = 14.03 + 0.324or ma = 13.624 kg/kg of fuelAir/fuel ratio = 13.624:1. - Theoretical air required
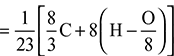
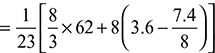 = 8.12 kg/kg of fuelExcess air supplied = 13.624 – 8.12 = 5.504 kg/kg of fuelPercentage of excess air supplied =
= 8.12 kg/kg of fuelExcess air supplied = 13.624 – 8.12 = 5.504 kg/kg of fuelPercentage of excess air supplied = 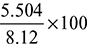 = 67.8%
= 67.8% - Flue gas analysis

Example 1.14
The mass analysis of a hydrocarbon fuel is as follows:
C = 84%, H2 = 5% and the balance is incombustible material. Find (a) the mass of air required per kg of fuel for complete combustion, (b) the analysis of the wet exhaust gases, by mass and volume, if 20 kg fuel is supplied, (c) the partial pressure of the steam formed in the exhaust gases if the total pressure of the exhaust gases is 103 kPa, (d) the heat carried away by dry exhaust gases formed per kg of fuel if the temperature of exhaust gas is 375°C and the ambient temperature is 24°C. Take cp for dry gases is 1.005 kJ/kgK.
[IES, 1988]
Solution
- Oxygen required for complete combustion of 1 kg of fuel
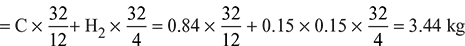 Air required per kg of fuel for complete combustion
Air required per kg of fuel for complete combustion
- The products of combustion per kg of fuel are:
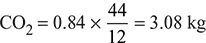
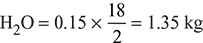 N2 = 14.96 × 0.77 = 11.52 kgWet products
N2 = 14.96 × 0.77 = 11.52 kgWet products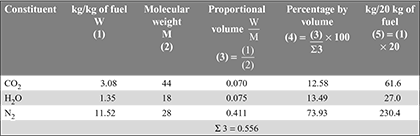
- Partial pressure of steam formed in the exhaust gases

- Mass of dry exhaust gases formed per kg of fuel= (mass of CO2 + mass of N2) per kg of fuel= 3.08 + 11.52 = 14.60 kgHeat carried away by dry exhaust gases per kg of fuel burned= 14.60 × 1.005 × (375 – 24) = 5150.223 kJ
Example 1.15
A sample of gaseous fuel has the following composition by volume:
H2 = 28%, CO = 10%, CH4 = 2%, CO2 = 16%, O2 = 2%, N2 = 42%.
If this fuel is burned with 50% excess air, determine (a) the volume of air per cubic metre of gas at the same temperature and pressure, (b) the volumetric analysis of dry products, (c) the mass of products per kg of fuel, (d) the mass of dry products per kg of fuel, and (e) the mass of air supplied per kg of fuel.
Solution
- Minimum volume of air required for complete combustion of 1 m3 of fuel,
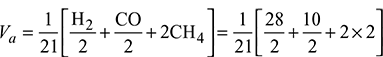 = 1.095 m3/m3 of gaseous fuelAir required with 50% excess air = 1.095 × 1.5 = 1.643 m3/m3 of gas
= 1.095 m3/m3 of gaseous fuelAir required with 50% excess air = 1.095 × 1.5 = 1.643 m3/m3 of gas - The constituents formed with 50% excess air are:CO2 = 0.16 + CO2 formed from CO + CO2 formed from CH4 = 0.16 + 0.10 × 1 + 0.02 × 1 = 0.28 m3N2 = 0.42 + N2 from air supplied = 0.42 + 1.643 × 0.79 = 1.718 m3O2 = 0.02 + O2 from excess air supplied = 0.02 + 1.095 × 0.5 × 0.21 = 0.135 m3Total dry flue gases formed = CO2 + N2 + O2 = 0.28 + 1.718 + 0.135 = 2.133 m3
-
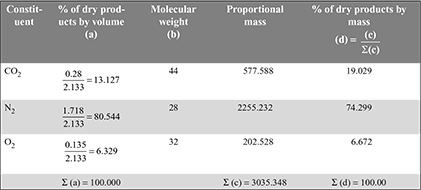
- Mass of air supplied per kg of fuel =
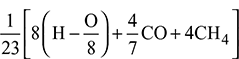
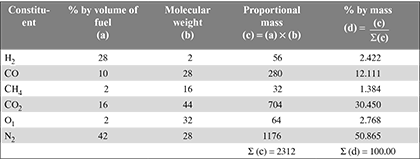
- Mass of air supplied =
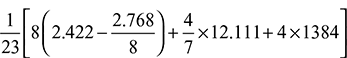 = 0.6318 kg/kg of fuelWith 50% excess air, mass of air supplied = 0.6318 × 1.5 = 0.9478 kg/kg of fuel
= 0.6318 kg/kg of fuelWith 50% excess air, mass of air supplied = 0.6318 × 1.5 = 0.9478 kg/kg of fuel
Example 1.16
A hydrocarbon fuel when burned with air gave the following Orsat analysis:
CO2 = 11.94%, O2 = 2.26%, CO = 0.41%, N2 = 85.39%
Determine (a) the air-fuel ratio on mass basis, (b) the percent of carbon and hydrogen in the fuel on mass basis, and (c) the percent of theoretical air supplied. Assume that air contains 21% oxygen by volume.
[IES, 1998]
Solution
Let 100x moles of air be used with fuel per 100 moles of dry exhaust products.
(aC + bH2)fuel + (21 × O2 + 79 × N2)air = 11.94 CO2 + 2.26 O2 + 0.41 CO + 85.39 N2 + dH2O
By molar balance, we have
Carbon: a = 11.94 + 0.41 = 12.35
Hydrogen: b = d
Oxygen: 21x = 11.94 + 2.26 +![]()
= 14.405 + 0.5d
Nitrogen: 79x = 85.39 or x = 1.08
From the above equation, we get
21 × 1.081 = 14.405 + 0.5d
d = b = 16.592
- Air-fuel ratio =
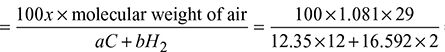 = 17.28
= 17.28 - % Carbon in fuel =
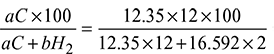 = 81.7%% Hydrogen in fuel =
= 81.7%% Hydrogen in fuel = 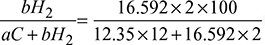 = 18.3%
= 18.3% - % Theoretical air supplied
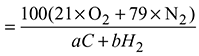 + (21 ×O2 + 79 × N2)
+ (21 ×O2 + 79 × N2) 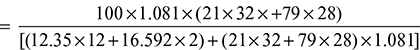 = 94.5%
= 94.5%
Example 1.17
The chemical formula for alcohol is C2H6O. Calculate the stoichiometric air/fuel ratio by mass and the percentage composition of the products of combustion per kg of C2H6O.
[GATE, 1998]
Solution
The chemical equation for complete combustion of given fuel can be written as follows:
C2H6O + 3O2 → 2CO2 + 3H2O
(1 × 46) + (3 × 32) = (2 × 44) + (3 × 18)
For complete combustion of 1 kg of C2H6O, oxygen required

= 2.087 × ![]() = 9.074 kg of air
= 9.074 kg of air
∴ A:F ratio for complete combustion = 9.074: 1
Also 46 kg of fuel produces products of combustion = 88 + 54 = 142 kg
∴ 1 kg of fuel produces ![]() = 3.087 kg of products of combustion
= 3.087 kg of products of combustion
(i.e., CO2 and H2O)
Hence, CO2 produced by fuel = ![]() × 100 = 1.913 or 191.3%
× 100 = 1.913 or 191.3%
H2O produced by fuel = ![]() × 100 = 1.174 or 117.4%
× 100 = 1.174 or 117.4%
Example 1.18
The products of combustion of an unknown hydrocarbon CxHy have the following composition as measured by an Orsat apparatus.
CO2 = 8.0%, CO = 0.9%, O2 = 8.8%, and N2 = 82.3%.
Determine (a) the composition of the fuel (b) the air-fuel ratio; and (c) the percentage of excess air used.
Solution
- Let a moles of oxygen by supplied per mole of fuel. The chemical reaction can be written as follows:CxHy + aO2 + 3.76 aN2 → 8CO + 0.9CO2 + 8.8O2 + 82.3H2 +
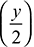 H2ONitrogen Balance gives 3.76a = 82.3or a = 21.89Oxygen Balance gives: 2a = 16 + 0.9 + 17.6 +
H2ONitrogen Balance gives 3.76a = 82.3or a = 21.89Oxygen Balance gives: 2a = 16 + 0.9 + 17.6 + 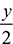 or y = 18.5Carbon balance givesx = 8 + 0.9 = 8.9Therefore, the chemical formula of the fuel is C8.9H18.5.The composition of the fuel is% Carbon =
or y = 18.5Carbon balance givesx = 8 + 0.9 = 8.9Therefore, the chemical formula of the fuel is C8.9H18.5.The composition of the fuel is% Carbon =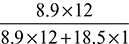 × 100 = 85.23%% Hydrogen = 14.77%
× 100 = 85.23%% Hydrogen = 14.77% - Air fuel ratio =
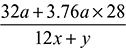
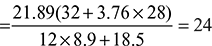
- % Excess air used
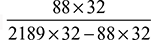 × 100 = 67.22%
× 100 = 67.22%
Example 1.19
The volumetric analysis of a gas is 14% CO2, 1% CO, 5% O2, and 80% N2. Calculate the fuel gas composition by mass.
Solution
Given: CO2 = 14% = 0.14 m3; CO = 1% = 0.01 m3; O2 = 5% = 0.05 m3; N2 = 80% = 0.8 m3
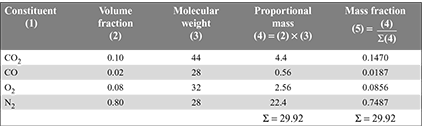
The volumetric analysis may be converted into mass analysis by completing the table as follows:
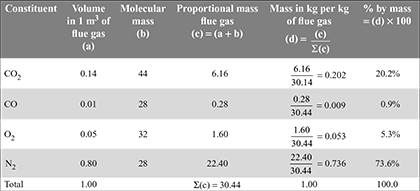
The fuel gas composition, by mass is given in the last column, i.e., CO2 = 20.2%, CO = 0.9%, O2 = 5.3%, and N2 = 73.6%.
Example 1.20
A blast furnace gas has the following volumetric composition:
CO2 = 11%, CO = 27%, H2 = 2%, and N2 = 60%.
Find the theoretical volume of air required for the complete combustion of 1 m3 of the gas. Find the percentage composition of dry flue gases by volume. Assume that air contains 21% of O2, and 79% of N2 by volume.
Solution
Given: CO2 = 11% = 0.11 m3, CO = 27% = 0.27 m3, H2 = 2% = 0.02% m3, N2 = 60% = 0.6 m3
We know that theoretical volume of air required
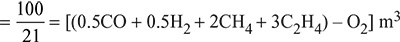
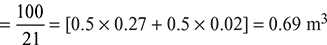
(∴ CH4, C2H and O2 are equal to zero)
We know that 1 m3 CO produces 1 m3 of CO2, therefore
Volume of CO2 obtained from the given 0.27 m3 of CO = 0.27 m3
and volume of CO2 already present in the fuel = 0.11 m3 (Given)
∴ Total volume of CO2 in the flue gas = 0.11 + 0.27 = 0.38 m3
Now volume of N2 from the theoretical air supplied = ![]() × 0.69 = 0.545 m3
× 0.69 = 0.545 m3
and volume of N2 already present in the fuel = 0.6 m3
∴ Total volume of N2 in the flue gas = 0.6 + 0.545 = 1.145 m3
and volume of the dry flue gas = Total volume of CO2 + Total volume of N2
= 0.38 + 1.145 = 1.525 m3
∴ Percentage of CO2 in the dry flue gas =![]() × 100 = 24.92%
× 100 = 24.92%
and percentage of N2 in the dry flue gas =![]() × 100 = 75.08%
× 100 = 75.08%
Example 1.21
The percentage composition by mass of a sample of coal as found by analysis is given as: C = 90%, H2 = 3.3%, O2 = 3.0%, N2 = 0.8%, S = 0.9%, and ash 2.0%.
Calculate the minimum mass of air required for the complete combustion of 1 kg of this fuel. If 50% excess air is supplied, find the total mass of dry flue gases per kg of fuel and the percentage composition of the dry flue gases by volume.
Solution
Given: C = 90% = 0.9 kg; H2 = 3.3% = 0.033 kg; O2 = 3% = 0.03 kg; N = 0.8% = 0.008 kg; S = 0.9% = 0.009 kg; Ash = 2% = 0.02 kg; Excess air supplied = 50%
We know that minimum mass of air required for complete combustion of 1 kg of fuel.
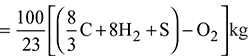
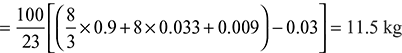
Total mass of dry flue gases per kg of fuel
Since 50% excess air is supplied, therefore actual amount of air supplied per kg of coal = 11.5 × 1.5 = 17.25 kg
∴ Excess air supplied = 17.25 – 11.5 = 5.75 kg
The products of combustion are represented by the following chemical equations:
C + O2 = CO2
2H2 + O2 = 2H2O
S + O2 = SO2
In addition to carbon dioxide, water, and sulphur dioxide, the excess oxygen and nitrogen will be available in the products of combustion. It may be noted that H2O (water vapour) is a wet gas, therefore, the dry flue gases are only carbon dioxide, sulphur dioxide, excess oxygen, and nitrogen. Let us now find the mass of each of these flue gases per kg of fuel.
We know that 1 kg of carbon produces 11/3 kg of carbon dioxide and 1 kg of sulphur produces 2 kg of sulphur dioxide.
∴ Mass of CO2 contained in 0.9 kg of carbon per kg of fuel = ![]() × 0.9 = 3.3 kg(i)
× 0.9 = 3.3 kg(i)
and mass of SO2 in 0.009 kg of sulphur per kg of fuel = 2 × 0.009 = 0.018 kg(ii)
We also know that the mass of excess O2 per kg of fuel = ![]() × Excess air supplied
× Excess air supplied
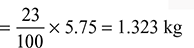
and mass of nitrogen in the products of combustion per kg of fuel

∴ Total mass of dry flue gases per kg of fuel = 3.3 + 0.018 + 1.323 + 13.283 kg
= 17.924 kg
First, let us find out the percentage composition of dry flue gases from the aforementioned data by mass. We know the following composition:
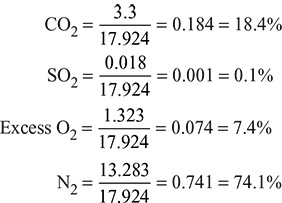
Now, let us convert this mass analysis of dry flue gases into volumetric analysis as follows:

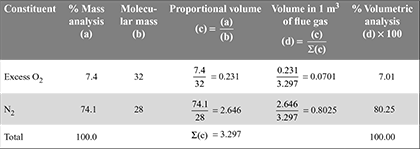
The percentage composition of dry flue gases (by volume) is given in last column, that is, CO2 = 12.68%, SO2 = 0.06%; Excess O2 = 7.01%; and N2 = 80.25%.
C = 85%, H2 = 12.5%, O2 = 2% and the residue 0.5%.
Example 1.22
A liquid fuel C7H16 is burned with 10% more air than the stoichiometric air. Assuming complete combustion, calculate (a) the mass of air supplied per kg of fuel and (b) the volumetric analysis of the dry products of combustion. Assume air contains 23% O2 by mass.
[IES, 2009]
Solution
- The chemical equation of combustion for the given fuel is:C7H16 + 11O2 = 7CO2 + 8H2O100 kg + 11 × 32 kg = 308 kg + 144 kg1 kg of fuel requires =
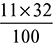 kg of O2
kg of O2 = 15.3 kg of air/kg of fuelWhen 10% more air than the stoichiometric air is burned, then actual mass of air supplied = 1.1 × 15.3 = 16.83 kg of air/kg of fuelMass of CO2 formed =
= 15.3 kg of air/kg of fuelWhen 10% more air than the stoichiometric air is burned, then actual mass of air supplied = 1.1 × 15.3 = 16.83 kg of air/kg of fuelMass of CO2 formed = 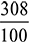 = 3.08 kg/kg of fuelO2 comes in exhaust from complete air supplied,N2 = 16.83 ×
= 3.08 kg/kg of fuelO2 comes in exhaust from complete air supplied,N2 = 16.83 × 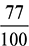 = 12.96 kg/kg of fuelO2 comes in exhaust from only excess air supplied,O2 = 15.3 × 0.1 ×
= 12.96 kg/kg of fuelO2 comes in exhaust from only excess air supplied,O2 = 15.3 × 0.1 × 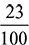 = 0.352 kg
= 0.352 kg - The mass is converted into volume as listed below in the table:
Example 1.23
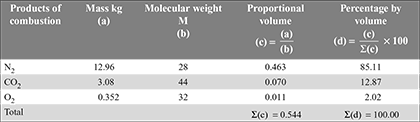
A four-cylinder engine of an automobile is converted to run on propane (C3H8) fuel. A dry analysis of engine exhaust gives volumetric percentage of CO, CO2 and O2, respectively at 9.79%, 4.90% and 2.45%. Write the resulting chemical reaction and find the equivalence ratio.
[IES, 2010]
Solution
Given: Volumetric analysis: CO = 9.7%, CO2 = 4.90%, O2 = 2.45%
The volumetric analysis of exhaust analysis can be converted to mass analysis as follow:
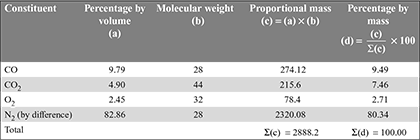
Percentage of C in C3H8 =![]() = 0.8182 or 81.82%
= 0.8182 or 81.82%
Percentage of H2 in C3H8 = 100 – 81.82 = 18.18%
Mass of air supplied per kg of fuel,
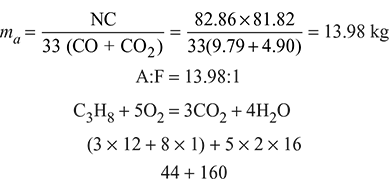
Oxygen required for complete combustion of 1 kg of fuel
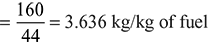
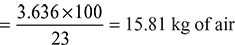
Minimum air required for complete combustion of fuel
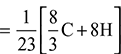
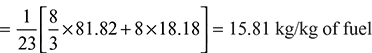

Example 1.24
Coal, having the following composition, is burnt in a furnace:
Carbon = 86.1%, hydrogen = 3.9%, oxygen = 1.4%, rest ash.
The volumetric analysis of dry products was as follows:
CO2 = 12.7%, CO = 1.4%, O2 = 4.1%, N2 = 81.8%. Determine the percentage of excess air. Determine the loss due to incomplete combustion, if CO burning to CO2 will release 24000 kJ/kg of carbon in CO.
[IAS, 1998]
Solution
Given: Composition of coal: C = 86.1%, H2 = 3.9%, O2 = 1.4%, Rest = 8.6% ash
Volumetric analysis of dry products:
CO2 = 12.7%, CO = 1.4%, O2 = 4.1%, N2 = 81.8%.
2CO + O2 → 2CO2 + 24,000 kJ/kg of C in CO
Mass of air supplied per kg of fuel, ![]()
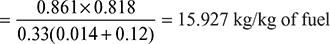
Theoretical air required for complete combustion of fuel
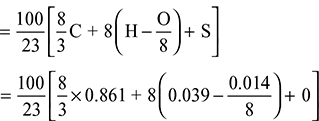
= 11.278 kg/kg of fuel
Excess air supplied = 15.927 – 11.278 = 4.649 kg/kg of fuel.
Carbon in fuel burned to ![]() = 0.10448 kg
= 0.10448 kg
Loss due to incomplete combustion = 0.10448 × 24000
= 2507.5 kJ/kg of fuel.
Example 1.25
The mass analysis of the petrol used in an engine was 85% C and 15% H2. The dry exhaust gas analysis showed that the percentage by volume of carbon dioxide was six times that of oxygen and that no carbon-monoxide was present. Calculate: (a) the air-fuel ratio by mass and (b) the percentage excess air supplied.
Assume air contains 23.2% O2 by mass or 20.9% O2 by volume.
[IAS, 1999]
Solution
Given: C = 0.85, H2 = 0.15% by mass
- Maximum mass of air required for complete combustion,
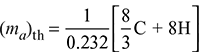
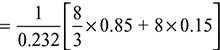 = 14.94 kgThe combustion equation for the given fuel can be written as:
= 14.94 kgThe combustion equation for the given fuel can be written as: where b, c, d and e are the products formed and ‘a’ is the amount of O2 supplied per kg of fuel.Balancing the mass of carbon from both sides, we haveb ×
where b, c, d and e are the products formed and ‘a’ is the amount of O2 supplied per kg of fuel.Balancing the mass of carbon from both sides, we haveb × 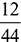 = 0.85 as 44 kg of CO2 contains 12 kg of carbon.b = 0.85 ×
= 0.85 as 44 kg of CO2 contains 12 kg of carbon.b = 0.85 × 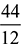 = 3.117 kgBalancing the mass of hydrogen from both sides, we havee ×
= 3.117 kgBalancing the mass of hydrogen from both sides, we havee × 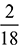 = 0.15 as 18 kg of H2O contains 2 kg of hydrogen.e = 1.35 kgBalancing the mass of O2 from both sides, we havea = b ×
= 0.15 as 18 kg of H2O contains 2 kg of hydrogen.e = 1.35 kgBalancing the mass of O2 from both sides, we havea = b × 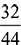 + c + e ×
+ c + e × 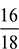 as 44 kg of CO2 contains 32 kg of O2 and 18 kg of water contains 16 kg of O2.a = 0.7273 b + c + 0.889 eNow balancing the mass of N2 from both sides, we havea ×
as 44 kg of CO2 contains 32 kg of O2 and 18 kg of water contains 16 kg of O2.a = 0.7273 b + c + 0.889 eNow balancing the mass of N2 from both sides, we havea × 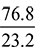 = d a = 0.302 dThus, we havea = 0.7273 × 3.117 + c + 0.889 × 1.35 = 2.267 + c + 1.2 = 3.467 + cVolumetric analysis of exhaust gases:
= d a = 0.302 dThus, we havea = 0.7273 × 3.117 + c + 0.889 × 1.35 = 2.267 + c + 1.2 = 3.467 + cVolumetric analysis of exhaust gases: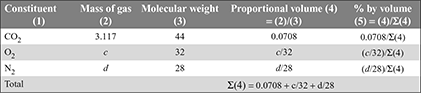 As per the given condition, percentage by volume of CO2 = 6 × percentage by volume of O2
As per the given condition, percentage by volume of CO2 = 6 × percentage by volume of O2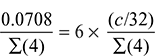 or 0.0708 = 6 ×
or 0.0708 = 6 × 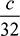 c = 0.3776 kg of O2 in the exhaust gases when one kg of fuel is burnt.We have a = 3.467 + 0.3776 = 3.8446 kg of O2 is supplied per kg of fuel.Amount of actual air supplied per kg of fuel, (ma)act = 3.8446 ×
c = 0.3776 kg of O2 in the exhaust gases when one kg of fuel is burnt.We have a = 3.467 + 0.3776 = 3.8446 kg of O2 is supplied per kg of fuel.Amount of actual air supplied per kg of fuel, (ma)act = 3.8446 × 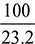 = 16.57 kg∴ A:F ratio = 16.57:1
= 16.57 kg∴ A:F ratio = 16.57:1 - % Excess air supplied =
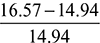 = 0.1091 or 10.91%
= 0.1091 or 10.91%
Example 1.26
A four cylinder engine of a truck has been converted to run on propane fuel. A dry analysis of the engine exhaust gives the following volumetric percentages:
CO2 = 4.90; CO = 9.79 and O2 = 2.45.
Calculate the equivalence ratio at which the engine is operating.
[IAS, 2001]
Solution
Given: Volumetric analysis of engine exhaust: CO2 = 4.90%, CO = 9.79%, O2 = 2.45%
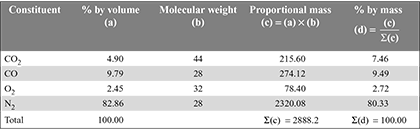
Mass of air supplied, ma = 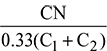
Propane: C5H12 + 😯2 = 5CO2 + 6H2O
Minimum amount of air required in kg per kg of fuel for complete combustion
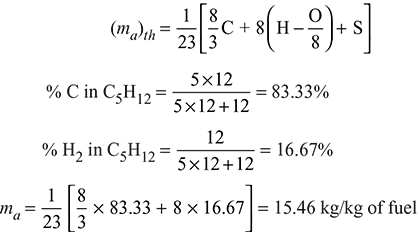
Actual A:F ratio = 15.46:1
Stoichiometric amount of air required per kg of fuel burned,
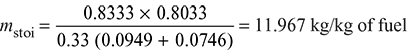

Leave a Reply