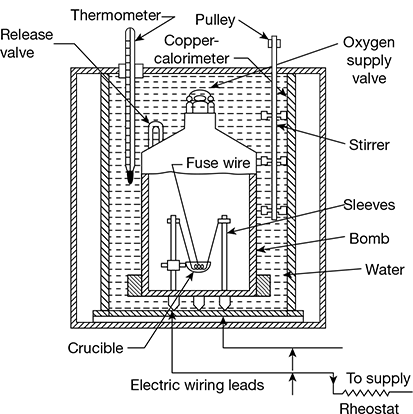
1 Construction
This is used to determine the higher calorific value of solid and liquid fuels. The bomb calorimeter shown in Fig. 1.2 consists of a strong steel shell, called the bomb, which can withstand a pressure of about 100 atm. The electric supply is provided at the bottom of the bomb. The silica or quartz crucible is supported on two pillars. It is also provided with a non-return and release valves. The bomb is placed in water bath and the water bath itself is placed in another container which acts as heat insulator. A thermometer and a stirrer are inserted in the outer vessel.
2 Working
A known quantity of fuel is taken in the crucible such that the fuse touches the fuel. The bomb is then charged with oxygen to a pressure of 30 bar. The oxygen charging non-return valve and release valve are closed tightly and the bomb is placed in water bath and closed with the cover. The water in the calorimeter is stirred and when the temperature remains steady, the fuel is ignited by passing a current through the fuse wire. The temperature of the bath starts increasing very quickly after the ignition and the readings on the thermometer are taken at regular intervals till the maximum temperature is reached.
The temperature gradually starts falling. When the temperature fall shows a steady rate, the readings are taken again for another five minutes to determine the cooling correction, as shown in Fig. 1.3. The cooling correction should be added to the measured temperature rise.
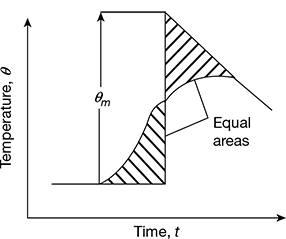
Figure 1.3 Temperature-time curve
Considering heat balance, we have
Heat given by the fuel due to combustion + Heat given by the combustion of fuse wire = Heat absorbed by the water and calorimeter.
mf × HCV + mfw × C.V. = (mw + ma)Δθcp
where mf = mass of fuel
mc = water equivalent of calorimeter
mw = mass of water in calorimeter
cp = specific heat of water
θm = recorded temperature rise
HCV = higher calorific value of fuel
C.V. = calorific value of fuse wire
∆θ = true temperature rise
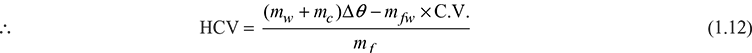
The water equivalent of calorimeter is determined by burning a fuel of known calorific value and using the aforementioned equation. The fuels used for this purpose are benzoic acid (C.V. = 111573 kJ/kg) and naphthalene (C.V. = 40690 kJ/kg).
3 Cooling Correction
The radiation correction is computed from Newton’s law of cooling. It states that the rate of heat loss due to radiation is proportional to temperature difference between the hot body and the surroundings. Thus,
δQ ∝ (θ2 − θ1)dt
The rate of temperature drop is determined from the temperature-time graph for the bomb calorimeter. Let it be r°C after the maximum temperature is reached. Therefore, during the period the temperature rises, it can be assumed that ![]() fall in temperature occurs due to radiation. If t is the time required for the maximum temperature to be reached, the temperature fall during that period due to radiation is
fall in temperature occurs due to radiation. If t is the time required for the maximum temperature to be reached, the temperature fall during that period due to radiation is ![]() Thus, for temperature fall,
Thus, for temperature fall,

Leave a Reply